
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
6th Edition
ISBN: 9780135765982
Author: Karen Timberlake, William Timberlake
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 17, Problem 87APP
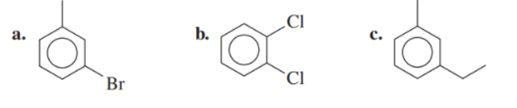
Name each of the following
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Q117
12.40 A compound called cinnamaldehyde is found in cinnamon.
Identify the functional groups in cinnamaldehyde. (12.1, 12.3)
CH3 CH — С -н
Cinnamaldehyde
Q 106
Chapter 17 Solutions
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
Ch. 17.1 - Prob. 1PPCh. 17.1 - Prob. 2PPCh. 17.1 - Prob. 3PPCh. 17.1 - Prob. 4PPCh. 17.1 - Prob. 5PPCh. 17.1 - Prob. 6PPCh. 17.1 - Prob. 7PPCh. 17.1 - Prob. 8PPCh. 17.1 - Prob. 9PPCh. 17.1 - Prob. 10PP
Ch. 17.1 - Prob. 11PPCh. 17.1 - Draw the condensed structural formula for each of...Ch. 17.1 - Prob. 13PPCh. 17.1 - Prob. 14PPCh. 17.1 - Prob. 15PPCh. 17.1 - Prob. 16PPCh. 17.1 - Prob. 17PPCh. 17.1 - Prob. 18PPCh. 17.2 - Prob. 19PPCh. 17.2 - Identify each of the following as an alkane,...Ch. 17.2 - Prob. 21PPCh. 17.2 - Prob. 22PPCh. 17.2 - Prob. 23PPCh. 17.2 - Prob. 24PPCh. 17.2 - Draw the condensed structural formula for the...Ch. 17.2 - Prob. 26PPCh. 17.2 - Prob. 27PPCh. 17.2 - Prob. 28PPCh. 17.2 - Prob. 29PPCh. 17.2 - Prob. 30PPCh. 17.2 - Prob. 31PPCh. 17.2 - Prob. 32PPCh. 17.3 - Prob. 33PPCh. 17.3 - Prob. 34PPCh. 17.3 - Prob. 35PPCh. 17.3 - Draw the line-angle formula for each of the...Ch. 17.4 - Prob. 37PPCh. 17.4 - Prob. 38PPCh. 17.4 - Prob. 39PPCh. 17.4 - Write the common name for each of the following:...Ch. 17.4 - Draw the condensed structural and line-angle...Ch. 17.4 - Draw the condensed structural and line-angle...Ch. 17.5 - Write the common name for each of the following:Ch. 17.5 - Write the common name for each of the following:Ch. 17.5 - Prob. 45PPCh. 17.5 - Prob. 46PPCh. 17.5 - Prob. 47PPCh. 17.5 - Draw the condensed structural formula for a and b...Ch. 17.6 - Prob. 49PPCh. 17.6 - Write the IUPAC and common name (if any) for each...Ch. 17.6 - Prob. 51PPCh. 17.6 - Prob. 52PPCh. 17.6 - Prob. 53PPCh. 17.6 - Prob. 54PPCh. 17.6 - Prob. 55PPCh. 17.6 - Prob. 56PPCh. 17.6 - Prob. 57PPCh. 17.6 - Write the IUPAC and common names, if any, for each...Ch. 17.6 - Draw the condensed structural formulas for a and b...Ch. 17.6 - Draw the condensed structural formulas for a and b...Ch. 17.7 - Write the common name for each of the following:...Ch. 17.7 - Prob. 62PPCh. 17.7 - Prob. 63PPCh. 17.7 - Prob. 64PPCh. 17.7 - Prob. 65PPCh. 17.7 - Prob. 66PPCh. 17.7 - Prob. 67PPCh. 17.7 - Prob. 68PPCh. 17.7 - Prob. 69PPCh. 17.7 - Prob. 70PPCh. 17.7 - Prob. 71PPCh. 17.7 - Prob. 72PPCh. 17 - The chapter sections to review are shown in...Ch. 17 - The chapter sections to review are shown in...Ch. 17 - Prob. 75UTCCh. 17 - Prob. 76UTCCh. 17 - Prob. 77APPCh. 17 - Prob. 78APPCh. 17 - Prob. 79APPCh. 17 - Prob. 80APPCh. 17 - Prob. 81APPCh. 17 - Prob. 82APPCh. 17 - Prob. 83APPCh. 17 - Prob. 84APPCh. 17 - Classify each of the following according to its...Ch. 17 - Classify each of the following according to its...Ch. 17 - Name each of the following aromatic compounds:...Ch. 17 - Prob. 88APPCh. 17 - Prob. 89APPCh. 17 - Draw the structural formula for each of the...Ch. 17 - Prob. 91APPCh. 17 - Prob. 92APPCh. 17 - Draw the condensed structural formula for each of...Ch. 17 - Draw the condensed structural formula for each of...Ch. 17 - Write the IUPAC name for each of the following:...Ch. 17 - Write the IUPAC name for each of the following:...Ch. 17 - Draw the condensed structural formulas for a and b...Ch. 17 - Prob. 98APPCh. 17 - Prob. 99APPCh. 17 - Prob. 100APPCh. 17 - Prob. 101APPCh. 17 - Draw the condensed structural formula for each of...Ch. 17 - Prob. 103APPCh. 17 - Prob. 104APPCh. 17 - Prob. 105CPCh. 17 - Prob. 106CPCh. 17 - The following problems are related to the topics...Ch. 17 - Prob. 108CPCh. 17 - The following problems are related to the topics...Ch. 17 - Prob. 110CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 12.60 Which of the following aldehydes or ketones are soluble in water? (12.3) |3| a. CH3-CH2 -C-CH3 b. CH3 — С —н CH3 c. CH3-CH2-CH-CH2-CH2-C-Harrow_forward12.38 The compound frambinone has the taste of raspberries and has been used in weight loss. Identify the functional groups in frambinone. (12.1, 12.3) Но CH,-CH,-C-–CH3 Frambinonearrow_forward12.58 Draw the condensed structural or line-angle formula, if cyclic, for each of the following: (12.3) a. formaldehyde c. 3-methyl-2-hexanone d. 3,5-dimethylhexanal b. 2-chlorobutanalarrow_forward
- 12.42 Which of the following will give a positive Tollens' test? (12.4) 1-propanol b. 2-propanol c. hexanal a.arrow_forwardSimple alcohols (just containing C's, H's and an OH, where the H on the O is the acidic H) have a pKa similar to water (15.7) hydronium ion (-2)arrow_forward(12.3)Which of the following has the strongest dispersion force between its molecules? O CH3CH3 O CH3CH₂CH₂CH₂CH3 O All of these have the dispersion forces with the same strength. O CH3CH₂CH₂CH3 O CH3CH₂CH₂CH₂CH₂CH3arrow_forward
- Please answer both the parts otherwise I'll downvotearrow_forward12.36 Avobenzone is a common ingredient in sunscreen. Its structural formula is shown. a. What functional groups are in avobenzone? b. What is the molecular formula and molar mass of avobenzone? c. If a bottle of sunscreen containing 236 mL has 3.0% (m/v) avobenzone, how many grams of avobenzone are present? Avobenzonearrow_forwardQ2. Using a series of steps, show how you would synthesize N-ethylpropanamide. You are given propanal and ethanamine to begin with. You may use any other reactants or catalysts that you need. The steps should be chemical reactions with the structures of the reactants and products drawn out. (10T)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY