
Interpretation:
To give the explanation whether the acid-catalyzed hydrolysis of acetamide is a reversible or irreversible reaction.
Concept introduction:
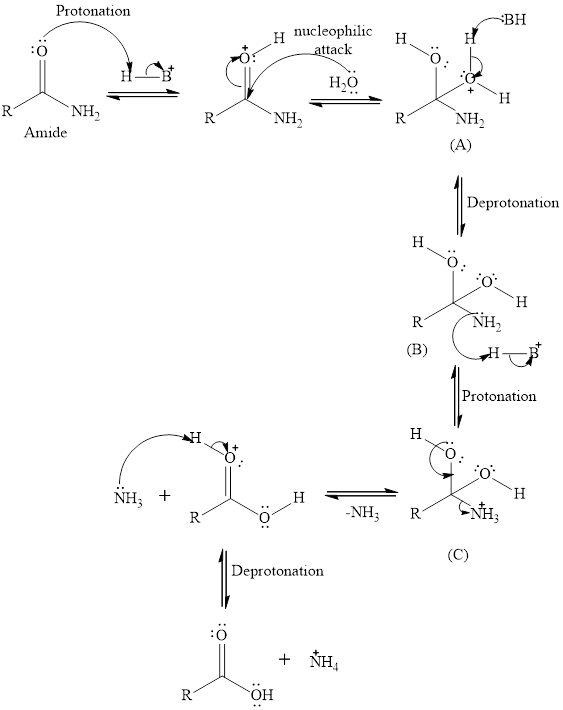
In acid hydrolysis of amide, acid protonates carbonyl oxygen of amide which increases the electrophilicity of the carbonyl carbon. Water acts as a nucleophile gets attached to the carbonyl carbon of amide to form tetrahedral intermediate(A) which on losing proton get converted to tetrahedral intermediate(B). The nitrogen of tetrahedral intermediate (B) gets protonated to form tetrahedral intermediate(C). The
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
EBK ORGANIC CHEMISTRY
- 18-29 Complete the equations for these acid—base reactions. (a) (b) (c) (d) (e)arrow_forwardSome penicillins cannot be administered orally because their B-lactam is rapidly hydrolyzed by the acidic environment of the stomach. What product is formed in the following hydrolysis reaction? H H H R H30* ČOOHarrow_forwardKetoprofen and ibuprofen both have a propanoic acid substituent. Explain why the identical subunits are synthesized in different ways.arrow_forward
- 2. Draw the structural formulas for the reactants and products for the following reactions. Name the organic product that forms D and Earrow_forwardCodeine can treat pain and cough but has a high risk for addiction and dependence. Which of the following statements about Codeine is false? H3C H- N-CH3 HO O The methoxy group masks an important binding group O The structure is converted to morphine in the brain O The structure is a weaker analgesic than morphine. O The structure acts as a prodrugarrow_forwardwhat products result when benzamide is treated with H2o, NaOH, and heat?arrow_forward
- describe how steric and electronic effects influence the postion of equilibrium when the electrophilic center of an aldehyde or ketone is under nucleophilic attack.arrow_forwardSome penicillins cannot be administered orally because their β-lactam is rapidly hydrolyzed by the acidic environment of the stomach. What product is formed in the following hydrolysis reaction?arrow_forwardDraw the molecules involved in the synthesis of aspirin. Is the forward reaction hydrolysis or condensation? Is the reverse reaction hydrolysis or condensation? Describe what occurs un each type of reaction with respect to acetylsalicylic acid.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





