
Concept explainers
(a)
Interpretation:
The
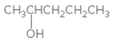
Concept Introduction:
When H2O is lost from a material it is called dehydration. When an alcohol is treated with a strong acid such as H2SO4, a water molecule is lost by breaking two adjacent bonds C-OH and C-H and forming a new double bond, producing an alkene. This is an elimination reaction.
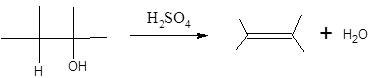
In this reaction more than one type of alkene may produce. But one of them is the major product.
According to Zaitsev rule, the major product alkene formed by elimination, is that which possess more alkyl groups bonded to it
(b)
Interpretation:
The alkene formed when the given alcohol is treated with H2SO4 should be determined. The major product should be predicted using Zaitsev rule.
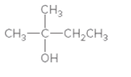
Concept Introduction:
When H2O is lost from a material it is called dehydration. When an alcohol is treated with a strong acid such as H2SO4, a water molecule is lost by breaking two adjacent bonds C-OH and C-H and forming a new double bond, producing an alkene. This is an elimination reaction.
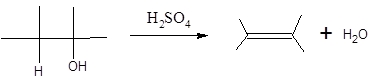
In this reaction more than one type of alkene may produce. But one of them is the major product.
According to Zaitsev rule, the major product alkene formed by elimination, is that which possess more alkyl groups bonded to it
(c)
Interpretation:
The alkene formed when the given alcohol is treated with H2SO4 should be determined. The major product should be predicted using Zaitsev rule.
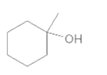
Concept Introduction:
When H2O is lost from a material it is called dehydration. When an alcohol is treated with a strong acid such as H2SO4, a water molecule is lost by breaking two adjacent bonds C-OH and C-H and forming a new double bond, producing an alkene. This is an elimination reaction.
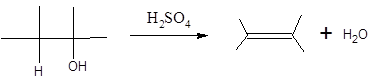
In this reaction more than one type of alkene may produce. But one of them is the major product.
According to Zaitsev rule, the major product alkene formed by elimination, is that which possess more alkyl groups bonded to it
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Draw the structures of the chief product formed when the following alcohols are dehydrated to alkenes: a. b.arrow_forwardExplain Addition of Alcohols—Acetal Formation ?arrow_forwardComplete the following syntheses – they may be two- or three-step processes. Include any necessary catalysts or reaction conditions. a) Prepare propanone from 1-propanolarrow_forward
- Name the following alcohol. O2-Bromo-4-ethylcyclopentanol 4-Ethyl-2-bromocyclopentanol O1-Ethyl-3-bromo-4-cyclopentanol O1-Bromo-4-ethyl-2-cyclopentanolarrow_forwardDraw and name ethers and heterocyclic ethers, including epoxides. Predict theirrelative boiling points, solubilities, and solvent properties.arrow_forwardAlkenes can be converted to alcohols by reaction with mercuric acetate to form a ß-hydroxyalkylmercury(II) acetate compound, a reaction called oxymercuration. Subsequent reduction with NaBH reduces the C-Hg bond to a C-H bond, forming the alkyl alcohol, a reaction called demercuration. Draw the structure of the Hg-containing compound and the final alcohol product formed in the reaction sequence. CH3 Hg(OOCCH3)2 NaBH4 HO™ Oxymercuration product Demercuration Product H₂O, THF Draw the alcohol product of demercuration. Draw the neutral product of oxymercuration. Omit byproducts. OH Hg Incorrectarrow_forward
- Explain Structure and Bonding of Alcohols, ethers, and epoxides ?arrow_forward1. 1-Octen-3-ol is a potent mosquito attractant commonly used in mosquito traps. A number of reactions, including hydrogenation, will transform 1-Octen-3-ol into a less effective molecule. What is a complete reaction equations for the hydrogenation of this alkenolarrow_forwardClassify each molecule as an aldehyde, ketone, or neither. AND Classify each molecule as an ester, ether, or neither.arrow_forward
- An alcohol does not react with aziridine unless an acid is present. Why is the acid necessary?arrow_forwardDraw the structural formula of the products formed when each alkene is treated with one equivalent of NBS in CH2Cl2 in the presence of light.arrow_forwardMany insects utilize cyclic ketal structures as pheromones such as the structure shown below. The biosynthetic pathway involves the cyclization of this acetal from the straight chain structure. Draw the straight chain structure that could be used to form this acetal. Use wedges and dashes to correctly depict the stereochemistry. H3C- >arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
