
Concept explainers
(a)
Interpretation:
The reactant alcohol should be identified when the
Concept Introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
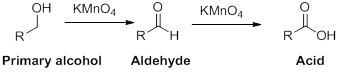
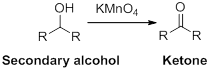
(b)
Interpretation:
The reactant alcohol should be identified when the aldehyde is the product.
Concept Introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
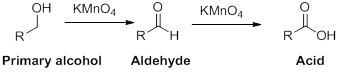
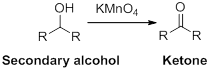
(c)
Interpretation:
The reactant alcohol should be identified when the acid is the product.
Concept Introduction:
Oxidation reaction:
Alcohol undergoes oxidation reaction using oxidising agent like
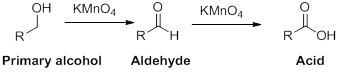
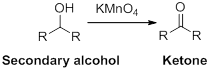
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
EP FUND.OF GENERAL,ORG...-MOD.MASTERING
- Salol, the phenyl ester of salicylic acid, is used as an intestinal antiseptic. Draw the structure of phenyl salicylate.arrow_forwardDraw a Lewis structure for hydroxylamine, NH2OH.arrow_forwardDrawn are four isomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?arrow_forward
- Draw the skeletal structure of the alkene that is needed as a starting material to prepare each of the following alcohols. Part 1 of 2 CH3CH(OH)CH3 Click and drag to start drawing a structure. ☑ :arrow_forwardWhat makes 2, 4-dinitrophenylhydrazine suitable for characterizing aldehydes and ketones? How would carbonyl groups in the two pheromones be distinguished by a suitable chemical method?arrow_forward2-butanol can be formed as the only product of the Markovnikov addition of H2O to two different alkenes. In contrast, 2-nonanol can be formed as the only product of the Markovnikov addition of H2O to just one alkene. To examine the difference, draw the alkene starting materials of each alcohol. Part 1 of 2 Draw the bond-line (skeletal) structures of the two alkene starting materials that can be used to synthesize 2-butanol via Markovnikov hydration. Click and drag to start drawing a structure. 0* Part 2 of 2 Draw the bond-line (skeletal) structure of the alkene starting material that can be used to synthesize 2-nonanol selectively via Markovnikov hydration. Click and drag to start drawing a structure.arrow_forward
- 2-butanol can be formed as the only product of the Markovnikov addition of H2O to two different alkenes. In contrast, 2-octanol can be formed as the only product of the Markovnikov addition of H2O to just one alkene. To examine the difference, draw the alkene starting materials of each alcohol. Draw the bond-line (skeletal) structures of the two alkene starting materials that can be used to synthesize 2-butanol via Markovnikov hydration. Part 1 of 2 Click and drag to start drawing a structure. : ☐ ☑ ⑤arrow_forwardDraw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic acidarrow_forwardFollowing are Fischer projections for a groupof five-carbon sugars, all of which are aldopentoses. Identify thepairs that are enantiomers and the pairs that are epimers. (Thesugars shown here are not all of the possible five-carbon sugars.)arrow_forward
- Following are Fischer projection for a group of five carbon sugars,all of which are aldopentoses. Identify the pairs that are enantiomers and the pairs that are epimers.(The sugar shown herebare not all of the possible five carbon sugars.)arrow_forwardFollowing are Fischer projections for a group of five-carbon sugars, all of which are aldopentoses. Identify the pairs that are enantiomers. CHO сно H-C- OH H-C-OH H-C- OH но-с — н н-с—он но- ČHOH ČH,OH сно CHO Н-с—он но—с— н H-C- OH H-C-OH но—с—н Н-с—он ČHOH ČH,OH сно сно н-с—он но—с —н но—с— н но -с — н H-C- OH но- C-H ČH,OH ČH,OHarrow_forwardDraw the structures of the following compounds. (Includes both new and old names.) 3-cyclopentylhexan-3-olarrow_forward
