
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.7C, Problem 12.3P
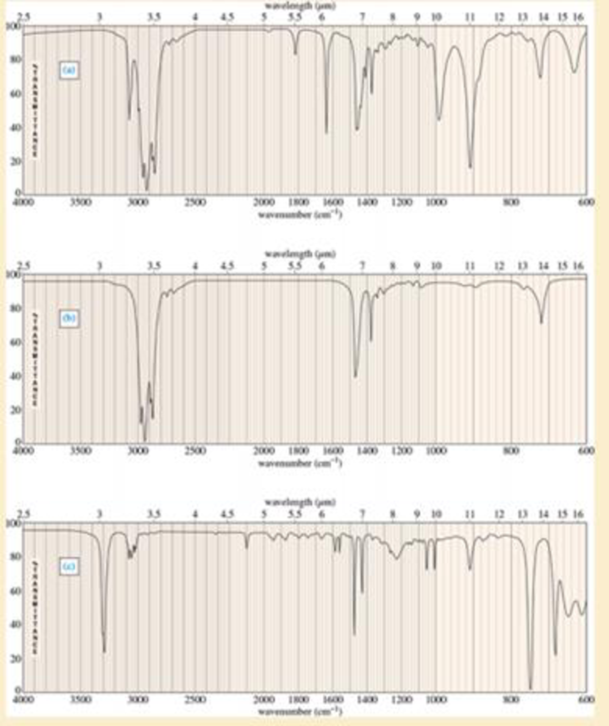
For each hydrocarbon spectrum, determine whether the compound is an
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Your classmate needs to decide if the spectrum shown below is a 13CNMR or an 1HNMR spectrum of 2-methyl propane. What is your advice to the student and why?
Which was likely lost to generate peak A?
a.)C
b.) CH2
c.) Br
d.)Cl
Based on the spectra you located, does your molecule have a carbonyl? If so, what functional group is it a part of (carboxylic acid, ketone, aldehyde, ester, amide) and what is the frequency (in wavenumbers) of the absorption peak? If not, what is the approximate frequency range for a carbonyl?
Does your molecule have either an –O-H or –N-H bond? If so, what functional group is it a part of (carboxylic acid, alcohol, amine, amide) and what is the frequency (in wavenumbers) of the absorption peak? If not, what are the approximate frequency ranges for an –O-H and an –N-H bond?
Does your molecule have either an alkyne or nitrile functional group? If so, which functional group is it and what is the frequency (in wavenumbers) of the absorption peak? If not, what is the approximate frequency range for a triple bond?
Chapter 12 Solutions
Organic Chemistry (9th Edition)
Ch. 12.3 - Complete the following conversion table. (cm1)...Ch. 12.5 - Which of the bonds shown in red are expected to...Ch. 12.7C - For each hydrocarbon spectrum, determine whether...Ch. 12.9A - Spectra are given for three compounds. Each...Ch. 12.10 - The infrared spectra for three compounds are...Ch. 12.12 - Prob. 12.6PCh. 12.14B - Identify which of these four mass spectra indicate...Ch. 12.15A - Show the fragmentation that accounts for the...Ch. 12.15A - Show the fragmentations that give rise to the...Ch. 12.15B - Ethers are not easily differentiated by their...
Ch. 12.15C - Prob. 12.11PCh. 12 - Prob. 12.12SPCh. 12 - Prob. 12.13SPCh. 12 - All of the following compounds absorb infrared...Ch. 12 - Prob. 12.15SPCh. 12 - Four infrared spectra are shown, corresponding to...Ch. 12 - Predict the masses and the structures of the most...Ch. 12 - Prob. 12.18SPCh. 12 - Prob. 12.19SPCh. 12 - (A true story) While organizing the undergraduate...Ch. 12 - Prob. 12.21SPCh. 12 - Prob. 12.22SPCh. 12 - An unknown, foul-smelling hydrocarbon gives the...Ch. 12 - covered a synthesis of alkynes by a double...Ch. 12 - Three IR spectra are shown, corresponding to three...Ch. 12 - Prob. 12.26SPCh. 12 - Prob. 12.27SPCh. 12 - Prob. 12.28SPCh. 12 - The ultimate test of fluency in MS and IR is...Ch. 12 - Prob. 12.30SPCh. 12 - Consider the following four structures, followed...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Indicate and circle the most important stretching signal from choices: N-H, O-H, C- - -N, C=O, C-O C8H10O2arrow_forwardWhat is M+, M+1 and M+2? Label these on spectrum. Calculate number carbons and nitrogen and using the equation. Start by assuming zero nitrogens; if the number of carbons does not come out to be a whole number, use the remainder to justify calculating number of nitrogens. This will not work out perfectly every time! Remember that these will be approximate depending on the signal intensity of the M+ peak and the presence of an [M-1]+ peak that can skew the isotope ratios. If this does not work out, use the value as an approximation to help guide your structure characterization. Calculate the number of oxygens using the equation. If there is no M+2 peak, state this. If the value is too low to be accurate, use it as an indicator for presence/absence of oxygen. This will not work out every time but will be used as a guide to determine chemical formula. Check the nitrogen rule – does it make sense with the M+ you observe and your calculations for N? Check whether fragments…arrow_forwardAnalyze the two spectra and find the peaks, shifts, and the kind of functional group the unknown is. The MW is 98.15 for this unknown.arrow_forward
- Label the major peaks, especially those those related to fumaric acidarrow_forwardWhat are the functional groups in this IR Spectra? Are there any that I am missing?arrow_forwardFirst picture: use the C13NMR spectrum below to determine the structural formula for the compound of molecular formula C5H10O Second picture: picture: use the HNMR spectrum below to determine the structural formula for the compound of molecular formula C6H12O2arrow_forward
- Indicate the multiplicity (singlet, doublet, triplet, quartet, quintet, sextet, septet, multiplet, etc.) of each signal in the HNMR spectrum of each of the following compounds:arrow_forwardWhat is the tR of peak A? What is the tR of peak B?arrow_forwardIdentify all of the peaks in the spectrum. State the Experimental wavelength, Literature wavelength, peak characteristic, and assignment. DOWN BELOW IS AN EXAMPLE... Experimental Wavelength (cm-1) Literature wavelength (cm-1) Peak characteristic assignment 3001cm-1, 3043cm-1 3010cm-1 sp2 C-H stretching alkene 2924cm-1 2900 -2980cm-1 sp3 C-H stretching Alkyl C-H 1614cm-1 1500 -1650cm-1 C=C stretch alkene 1443cm-1 1350-1450cm-1 C-H bend C-Harrow_forward
- How do I interpret, label, and determine which molecule this H NMR graph represents? The three molecule choices are methylene cyclohexane, 3-methylcyclohexene, or 1-methylcyclohexene.arrow_forwardMy peak at about 3500 cm-1 is facing upwards, what could be the cause? There is supposed to be peak at 1600 for C=C group but its only small, why? Context: Used KBr pellets, compound is supposed to be cyclohexenearrow_forwardSolve correctly please. Q.For each image, please find what's the 13C splitting pattern w/ the peak from the carbon circled in red. Assign each letter to the following answer choices: singlet, doublet, triplet, quartet or quintet. Please explain, show step by step:arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY