
Concept explainers
(a)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidation of primary alcohol to
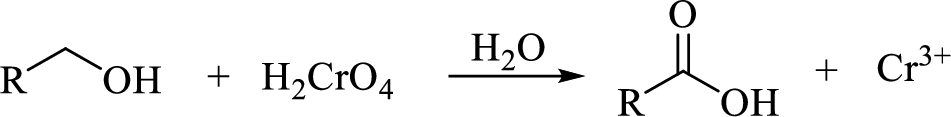
(b)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Reaction with
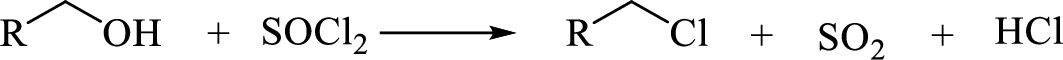
(c)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Reaction with
Secondary alcohols may react by

(d)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Reaction with
Secondary alcohols may react by

(e)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidation of primary alcohol to carboxylic acid: A primary alcohol is oxidized to a carboxylic acid by chromic acid. The mechanism involves initial formation of an alkyl chromate intermediate, followed by reaction with base to remove a proton, generating the carbonyl group of an aldehyde and simultaneously reducing the chromium (VI) to chromium (IV). An initially formed aldehyde adds water, generating an aldehyde hydrate, which is oxidized according to the same mechanism to give the carboxylic acid.
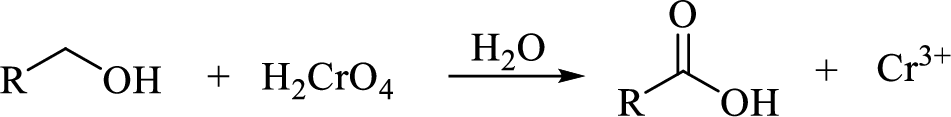
(f)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidative cleavage of a glycol:
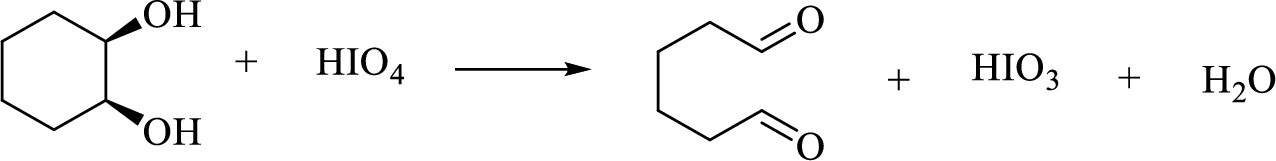
(g)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidative cleavage of a glycol:
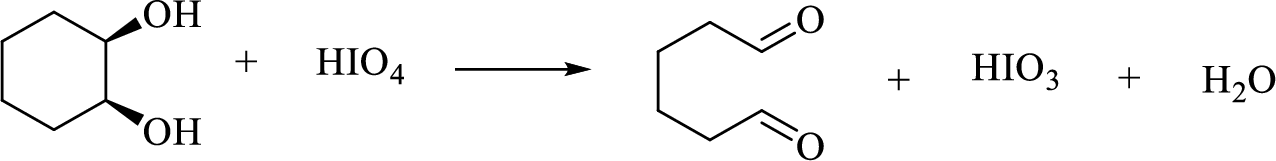
(h)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Reaction with
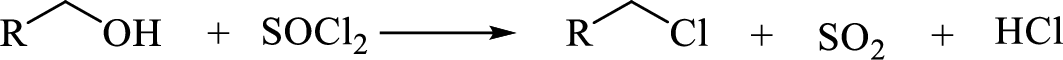
(i)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidation of primary alcohol to an aldehyde: The oxidation of a primary alcohol to an aldehyde can be carried out using pyridinium chlorochromate (PCC). Because there is no water, the aldehyde does not form the hydrate, and the oxidation reaction stops at the aldehyde stage. Alternatively, Swern or Dess-martin oxidation can be used.

(j)
Interpretation:
The major product of each of the given reaction has to be drawn.
Concept Introduction:
Oxidation of primary alcohol to an aldehyde: The oxidation of a primary alcohol to an aldehyde can be carried out using pyridinium chlorochromate (PCC). Because there is no water, the aldehyde does not form the hydrate, and the oxidation reaction stops at the aldehyde stage. Alternatively, Swern or Dess-martin oxidation can be used.

Trending nowThis is a popular solution!

Chapter 10 Solutions
OWL V2 with MindTap Reader and Student Solutions Manual eBook for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Aldehydes and ketones react with one molecule of an alcohol to form compounds called hemiacetals, in which there is one hydroxyl group and one ether-like group. Reaction of a hemiacetal with a second molecule of alcohol gives an acetal and a molecule of water. We study this reaction in Chapter 16. Draw structural formulas for the hemiacetal and acetal formed from these reagents. The stoichiometry of each reaction is given in the problem.arrow_forwardPhenols are aromatic rings with an alcohol functional group attached directly to the ring. These compounds have unique acidity and solubility for alcohol groups. Predict the solubility of this phenol in water.arrow_forwardComplete the following reaction by drawing structural formulas for the organic product(s). CH₂ ondeloom CH₂C CH₂ + H₂O H*arrow_forward
- Aldehydes and ketones are soluble in organic solvents True False The IUPAC name of the compound CH3CH2COCH3 is ******** The reaction between strong base and C6H6Cl is called The reaction between phenol and nitric acid product O-nitro phenol p-nitro phenol picric acid 2,4-nitrophenol إجابتك إجابتكarrow_forwardWhen trans-2-chloro-1-cyclohexanol is treated with a base, cyclohexene oxide is the product. However, when cis-2-chloro-1-cyclohexanol is treated with a base, the product is cyclohexanone A.Write the equation for the reaction between trans-2-chloro-1-cyclohexanol and the base to yield the cyclohexene oxide B.Why doesn’t the cis isomer yield the oxide? C.Write the mechanism for each of the two reactions.arrow_forwardUsing the relationship between the structure and the boiling point. Obtain a table for the listed chemicals below showing how the boiling point will be different with (1) the different chemical structures, (2) with different functional groups. Boiling Point and Functionalized Compounds: Chemical: Cyclohexanol Cyclohexanone Cyclohexene Benzaldehyde Benzyl alcoholarrow_forward
- Ethers can be classified as open chain or cyclic. Open chain ethers can contain rings. What is the structural difference between open chain ethers that contain rings and cyclic ethers?arrow_forwardDraw structural formulas for the alkene that gives each alcohol upon hydroboration-oxidation.arrow_forward1. 1-Octen-3-ol is a potent mosquito attractant commonly used in mosquito traps. A number of reactions, including hydrogenation, will transform 1-Octen-3-ol into a less effective molecule. What is a complete reaction equations for the hydrogenation of this alkenolarrow_forward
- What structural and/or molecular properties of alcohols contributes to their reactivity with oxidizing reagent?arrow_forwardenumerate the properties of alcohols contributing to their reactivity with an oxidizing reagent?arrow_forwardProvide the major organic product of the following reaction.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





