
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Transcribed Image Text:A cvg.cengagenow.com/ilrn/takeAssignment/takeCXPCompliantActivity.do?locator%=Dassignment-take
Ogevious Page 3 of 5
Next O
References
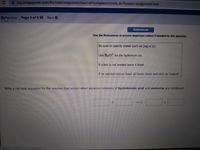
Use the References to access important values if needed for this question.
Be sure to specify states such as (aq) or (s).
Use H3 0 for the hydronium ion.
If a box is not needed leave it blank.
If no reaction occurs leave all boxes blank and click on "submit".
Write a net ionic equation for the reaction that occurs when aqueous solutions of hydrobromic acid and ammonia are combined.
(als-asp/take
Expert Solution
arrow_forward
Step 1
- Write the balanced molecular equation
- Split electrolytes to their corresponding ions to achieve complete ionic equation
- Cancel out common ions from both sides of complete ionic equation to achieve the net ionic equation
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A precipitate forms when aqueous solutions of chromium(II) iodide and potassium carbonate are combined.arrow_forwardWhat is the difference between an aqueous solution with dissolved ions within it and a solution that has a precipitating reaction occurring?arrow_forwardWrite the balanced molecular chemical equation for the reaction in aqueous solution for magnesium chloride and silver nitrate. If no reaction occurs, simply write only NR.arrow_forward
- Write the net ionic equation for the reaction that occurs, if any, when solutions of aluminum nitrate and sodium hydroxide are combined, producing an insoluble precipitate. What is/are the products?arrow_forwardWhat is the net ionic equation when hydrobromic acid reacts with potassium hydroxide ?arrow_forwardWrite a net ionic equation for the reaction that occurs when aqueous solutions of hydrobromic acid and potassium hydroxide are combined.arrow_forward
- write a net ionic equation for the reaction that occurs when manganese (II) suflide and excess hydrochloric acid (aq) are combinedarrow_forwardWrite the net iconic equation, if any, that occurs when aqueous solutions of: silver nitrate and ammonium carbonate are mixed.arrow_forwardWhat are the spectator ions present when aqueous solutions of rubidium phosphate and yttrium(II) bromide are mixed?arrow_forward
- Write the balanced neutralization reaction that occurs between sodium hydroxide and acetic acid.arrow_forwardWrite the balanced COMPLETE ionic equation for the reaction when iron(III) chloride and sodium sulfide are mixed in aqueous solution.arrow_forwardWrite a net ionic equation for the reaction that occurs when aqueous solutions of ammonia and hydrobromic acid are combinedarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY