Q: Use the dropdown menus in the sentence below to indicate if Ha and Hb within the structure are homot...
A: Concept: Homotopic hydrogen: In any molecule two hydrogens called as homotopic if replacing either ...
Q: Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its r...
A: Molarity = Number of moles of compound / Volume of solution (L)
Q: Write molecular equation for the following precipitation reaction. Iron(II) chloride and sodium sulf...
A:
Q: What mass in grams of NaCl would need to be added to 2551 g of water to increase the boiling tempera...
A:
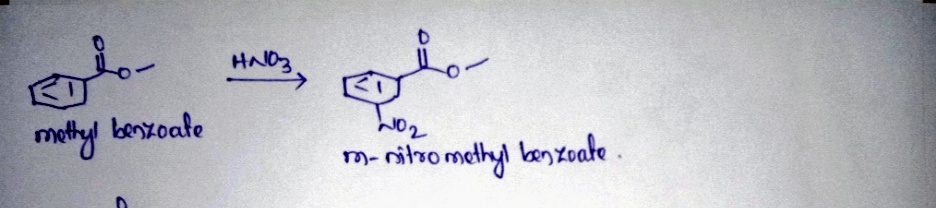
Q: 3-nitrobenzoate
A: 1HNMR spectrum of Methyl Benzoate has 4 peaks observed, but in the case of Meta Nitro Methyl Benzoat...
Q: Liquid octane (CH3(CH2)6CH3) will react with gaseous oxygen (O2) to produce gaseous carbon dioxide (...
A:
Q: Write a balanced chemical equation which corresponds to the following equilibrium constant expressio...
A:
Q: 24. Which of the following is the 1,4-addition product of Brz to the molecule given below? Br Br Br ...
A:
Q: In sigma MO the electron density is concentrated around a line between the two nuclei of the bonding...
A: Molecular orbital can be defined as the orbital formed by addition or subtraction of atomic orbitals...
Q: Find the r
A: Ptotal = P (CH3CHO) + P (CH4) + P(CO) Also, P(CH4) = P (CO) So, Ptotal = P (CH3CHO) + 2 P (CH4) Now...
Q: The hydronium ion concentration in a solution of an unknown weak acid is 5.77 x 1011. What is the pK...
A: Pka value is depends upon concentration of H+ ions in the solution.
Q: concentration in serum taken from a patient being treated with litnium for ressive illneSS wa sing f...
A: Concentration is the measure of how much of a given substance is mixed with another substance.
Q: Which of the following sets of atomic orbitals will form a molecule with the longest bond length? * ...
A: The atomic orbitals forming a molecule with the longest bond length = ?
Q: a Write the molecular equation which completes the following. The reaction occurs in an aqueous solu...
A: In balanced reaction number of each atom on reactant side is equal to number of each atom on product...
Q: Li + K2SO4 ----------> Li2SO4 + 2 K How many grams of potassium are produced if 3 moles of K2SO4 ar...
A: Given : We have to calculate the mass of K produced.
Q: The energy penalty that is accrued when large groups in a molecule are forced into proximity to one ...
A: This question clearly discusses about the energy associated with positional effect of substitutions ...
Q: Water sample analysis is conducted using AAS, the result shows that the absorption of sodium (Na) io...
A: A question based analytical process that is to be accomplished.
Q: Select which of the following is not a feature of carrier gas used in gas chromatography? A. It must...
A: Gas Chromatography is a technique used for separating vaporised compounds without their decompositio...
Q: Draw the product of this reaction. Ignore inorganic byproducts. KMNO4, NaOH cold Select to Draw
A:
Q: construct and show the structural formula of Glucose, Fructose, Ammonia, Ethanol, Acetic Acid
A: Draw and write structure formula of the given --
Q: How many grams of CH liquid must decompose according to the following chemical equation to transfer ...
A: The given thermochemical equation is: C6H6(l) ---> 3C2H2(g) ; ∆H = 630 kJ Given the required a...
Q: Draw the structure 3,5-diethyl-3-methyloctane in the window below.
A:
Q: Hydrolysis of an ester produced the two organic products shown. What is the structure of the origina...
A: Esters hydrolyse in acidic media to form alcohol and acid while in alkaline media to form alcohol an...
Q: What was the concentration of my stock solution if I used 26mL of it to make 116ml of a 1.5 M diluti...
A: Given that, V1= 26 mL , M1=? V2=116 mL , M2= 1.5 M
Q: Compute for the required amount of the ingredients necessary to make 20g suppository. The photo sho...
A: Given that : We have to compute for the required amount of the ingredients necessary to make 20g sup...
Q: A student used 1.50 g of manganese to react with excess fluorine to form manganese fluoride. If 4.09...
A:
Q: 12. Rank order the following carboxylic acids from strongest acid to weakest acid: COOH COOH (i) (ii...
A: An acid is called strong acid when it's conjugate base after deprotonation is most stable ,here ele...
Q: FOR PRACTICE 15.2 Consider the equation: CHCI3(8) + Cl2(8) CCI4(8) + HCI(g) The initial rate of reac...
A:
Q: Express the rate of the following reaction equation in terms of the rate of concentration change for...
A: Given :- 2SO2(g) + O2(g) → 2SO3(g) To write :- rate of reaction in terms of concentration change fo...
Q: What type of bond is the C-F bond in the nonpolar molecule carbon tetrafluoride (CF4)? polar ionic i...
A: Given, Non polar molecule : CF4
Q: Select the compound that would show a larger than usual M + 2 peak. O A. CH3CH2F O B. CH3CH2CH2| OC....
A: Concept: Effect of isotope in mass spectrometry: Mass spectrometry distinguishes elements based on ...
Q: 14.48 A compound (C3H10O) has the IR and 'H NMR spectra presented in O Figure 14.51. What is its str...
A: The structure of the following is:
Q: 1. Select the incorrect statement. q is the symbol for heat q = mC;AT The unit for g is Joules q is ...
A:
Q: The diol shown undergoes reactions with reducing and oxidizing agents to yield two different product...
A: Given : We have to draw the product for the given reactants.
Q: Calculate the molality of a solution formed by adding 6.832 g of methanol (CH3OH) to 100.00 mL of wa...
A: Molality is defined as number of moles of solute dissolved per kilogram of solvent.
Q: Consider the following chemical equilibrium: C(s) +2 H, (g) =CH, (g) Now write an equation below tha...
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical eq...
Q: Plastic sulfur is composed of a) S8 molecules c) S2 dimers O b) Sx polymers
A: The plastic sulfur is also called as x-sulfur.
Q: Differentiate between gel filtration and gel permeation chromatography.
A: Given : We have to tell what is the difference between gel filtration and gel permeation method.
Q: What is a calibration curve? (select the BEST description) O a. A calibration curve is a serial dilu...
A: As you have asked two questions, I will give you the answer for the first one.
Q: 16. Which of the intermediates shown below is generated during the following reaction? H,SO4 OH OCH2...
A:
Q: Select the IUPAC name for the following structure: CH3 CH-CH, CH,CH, CH,-CH-CH-CH,-CH-CH, CH,-C-CH, ...
A: The compound given is,
Q: 1. What produces the dry sensation in your mouth when drinking wine? 2. What happens when tannins co...
A: Human saliva is full of protein this makes it so slippery. When we drink wine the tannins and sa...
Q: propose a retrosynthesis, and a way in which this diene can be synthesized
A: Retrosynthesis of 1,3-butadiene.
Q: Use Beer’s Law to calculate the concentration of a sample of cyanmethaemoglobin if it has an absorba...
A:
Q: Retention time can be used to identify a compound in a mixture using gas chromatography. Choose a pa...
A: We have to tall which parameter will not affect the retention time of a compound in a gas chromatogr...
Q: How do I calculate the standard deviation of background (baseline) on the UV-Vis absorbance measurem...
A:
Q: Explain why carbon is such a unique atom. Include a discussion about molecular diversity of carbon-b...
A: Carbon has four electrons in the outer most shell.It has electronic configuration of 1s2 2s2 2p2, it...
Q: How many Chlorine atoms are there in phosgene?
A: Phosgene is a colorless gas having molar mass equals to 98.92 g/mol. It is a suffocating gas.
Q: he imaginary element A has molar mass of 31.0 g/mol and the imaginary element X has molar mass of 24...
A: Chemical equation is symbolic representation of chemical reaction, in this equation reactants writte...
Q: If the pKa of water is 15.7 and the pKa of HF is 3.2, which is the stronger base, OH or –F? -F -OH
A: Dissociation of a weak acid is given by: HA ⇌ H+ + A- The acid dissociation constant of this aci...
Would you expect a change in the frequency for the ester stretch between starting material and product in the nitration of methy benzoate?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- How you would test the relative reactivities of 1-butanol vs. 2-butanol in a nucleophilic substitution with sodium bromideWhat is the difference between activated pyridine and unactivated pyridinewhat would a detailed step-by-step mechanism be for a reaction between bromine and trans-stilbene if the solvent, ethanol, is a stronger nucleophile than the bromide ions?
- Why should you keep your head out of the fume hood during the reaction between tetraphylcyclopentadienone and Dimethyl acetylenedicarboxylate? Because the addition of heat to this experiment increases the pressure within the reaction vessel, the apparatus could explode unexpectedly. O Due to the formation of carbon monoxide gas, which is toxic if inhaled. Due to the formation of carbon monoxide, carbon dioxide, and ethylene gas, which are all toxic when inhaled. O All of the aboveWhat change can be made to allow for a faster rate of reaction if poor solubility of KOH in ethanol slowed the rate and led to slow deprotonation of dibenzyl ketone?Explain how you would test the relative reactivities of 1‐butanol vs. 2‐butanol in a nucleophilic substitution with sodium bromide.
- What mechanism does the reaction between benzyl chloride and triphenylphosphine go by? A lower yield of phosphonium salt is obtained in refluxing benzene than in xylene. Look up the boiling points for these solvents and explain why the difference in boiling points might influence of the yield? Why are the starting materials soluble in xylene but the product (phosphonium salt) is not soluble in xylene? We use petroleum ether to wash the phosphonium salt in filtration. Can we use water instead? Why?Please show the steps of the separation of a mixture of methyl phenyl ketone and N-methyl amine and indicate the results observed after each step.Briefly explain why OtBu- sometimes favored over hydroxide as an elimination reagent?
- The base-promoted rearrangement of an -haloketone to a carboxylic acid, known as the Favorskii rearrangement, is illustrated by the conversion of 2-chlorocyclohexanone to cyclopentanecarboxylic acid. It is proposed that NaOH first converts the a-haloketone to the substituted cyclopropanone shown in brackets and then to the sodium salt of cyclopentanecarboxylic acid. (a) Propose a mechanism for base-promoted conversion of 2-chlorocyclohexanone to the proposed intermediate. (b) Propose a mechanism for base-promoted conversion of the proposed intermediate to sodium cyclopentanecarboxylate.Why is the bromination (EAS) of phenyl ester slower than phenol?What is the theoretical yield of an aldol condensation reaction involving cyclopentanone as the ketone and p-tolualdehyde as the aldehyde using sodium hydroxide as the base given the following quantities? Mass of Cyclopentanone used (0.2 mL): 0.2 g, Mass of p-Tolualdehyde used (0.8 mL): 1 g, Volume of 2 M sodium hydroxide: 3mL (excess).

