
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Why are the major structures the ones where carbon has incomplete octets? Isn't the first rule for major resonance structures that the structure have as many full octets as possible?
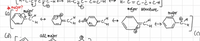
Transcribed Image Text:C=c-H
H- C= ċ-ċ =CH
t major?
magor strcture
major
major
ンH
H.
۲ )ف(
(c)
all major
J:0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Question: Consider a hypothetical three-atom molecule, XYZ, with atoms X and Z having a partial positive charge and atom Y having a partial negative charge. The molecule exhibits resonance, and its resonance structures are in equilibrium. Given that the bond lengths between the atoms are shorter than the sum of their van der Waals radii, explain the possible factors that contribute to this phenomenon and the implications of this bond length on the stability and reactivity of the molecule XYZ.arrow_forwardHow many equivalent resonance structures that follow the octet rule can be drawn for O3? One Two Three O Four This molecule has no resonance structures.arrow_forwardDraw a Lewis structure for the following ions/molecules, including any resonance structures and/or formal charges. Place a box around any contributing resonance structures XeO4^-2, BrF5, CHF2CL What is the electron group geometry around the central atom for each of the above ions/molecules? For the above ions/molecules, what is the shape of the entire molecule/ion of the entire ion/molecule? Identify polar bonds with dipole arrows Indicate whether each of the ions/molecules is POLAR or NON-POLARarrow_forward
- Predicting the single-bonded molecular compounds formed by two elements.arrow_forwardExplain why acetic acid does not have any resonance structures while the acetate ion does ? How does this affect the bond order of these molecules ?arrow_forwardConsider the SO 3 molecule. How many valence electrons does the molecule have? How many lone pairs of electrons are on the central atom? How many lone pairs of electrons in total are on the substituent atoms? How many resonance structures can be drawn for the molecule? If the ion doesn't exhibit resonance, indicate "1" as only one structure can be drawn for the molecule.arrow_forward
- Which of the following molecules contains at least one atom that has an expanded (hypervalent) octet?arrow_forwardWhy do sodium ion and choride ion form a compound? A) The energy of the formation of the negative chloride is overcome by the energy of the formation of the positive sodium ion. B) The lattice energy between the positive sodium and the negative chloride ions overcome the endothermic formation of the ions C)The energy of the formation of the positive sodium ion is overcome by the energy of the formation of the negative chloride ion. D) The energy of the formation of the positive sodium ion is canceled by the formation of the negative chloride ion.arrow_forwardDeciding whether a Lewis structure satisfies the octet rule Decide whether these proposed Lewis structures are reasonable. proposed Lewis structure Is the proposed Lewis structure reasonable? O Yes. O No, it has the wrong number of valence electrons. The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. fä=ö The correct number is: No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* O Yes. No it hae the wrong number of valence olactrons Explanation Check O 2021 McGrn P Type here to search Esc FnLk F1 F2 F3 F4 F5 F6 F7 F8 F9 %23 $4 3 6. 7 8. W E T Y Tab S ( CO LL :Z : : 0arrow_forward
- Consider the following ion: BrO3−. a) Show the full electron configuration for Br. b) Draw the most correct Lewis structure for BrO3− and briefly explain why your Lewis structure is correct. c) If the structure is stabilised by resonance, draw at least one of the possible resonance forms. If it is not stabilised by resonance, briefly explain why. d) What is the electronic geometry of BrO3−? What is its molecular shape? e) Does BrO3− have a dipole moment? Briefly justify your answer. f) On average, would you expect IO3− to have longer or shorter bonds than BrO3−? Briefly explain your answer. g) Which of the following molecules would you expect to have the lowest vapour pressure? Briefly explain your choice. (IMAGE WITH POSSIBILITIES) h) What is the molecular formula for Compound C? What is the empirical formula for Compound C?arrow_forwarddiagram of a molecule that shows atoms based on their a periodic table symbols. These symbols are surrounded by pairs of dots to show valence electrons that could be lone pairs or bonding pairs?arrow_forwardUse Lewis theory to determine the formula for the compound that forms between each of the following pairs of elements. Ca and Te Express your answer as a chemical formula. Mg and Br Express your answer as a chemical formula. Na and S Express your answer as a chemical formula. In and O Express your answer as a chemical formula.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY