
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
1) An ideal gas in a sealed container has an initial volume of 2.80L. At constant pressure, it is cooled to 18.00 Celsius, where it’s final volume is 1.75L. What was the initial temperature in Celsius?
2)What volume of H2O is produced when 6.30 mol of C2H4 reacts with oxygen gas at STP?
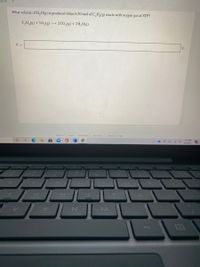
Transcribed Image Text:of 10
What volume of H, O(g) is produced when 6.30 mol of C, H,(g) reacts with oxygen gas at STP?
C,H,(g) +30,(g) 2CO,(g) +2H,0(g)
V =
about us careers privacy policy tems of use contact us help
351 PM
H
M
Alt
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Sometimes in lab we collect the gas formed by a chemical reaction over water (see sketch at right). This makes it easy to isolate and measure the amount of gas produced. Suppose the O₂ gas evolved by a certain chemical reaction taking place at 30.0 °C is collected over water, using an apparatus something like that in the sketch, and the final volume of gas in the collection tube is measured to be 114. mL. g x10 X collected Ś gas Sketch of a gas-collection apparatus Calculate the mass of O₂ that is in the collection tube. Round your answer to 2 significant digits. You can make any normal and reasonable assumption about the 2 reaction conditions and the nature of the gases. water IS chemical reactionarrow_forwardA mixture of solid calcium oxide, CaO, and solid barium oxide, BaO, weighing 3.145 g is added to a 2.00 L flask containing carbon dioxide, CO2, at 30 ∘C. The CaO and BaO react completely with the carbon dioxide gas to form solid CaCO3 and BaCO3, respectively. Calculate the moles of CO2 consumed by the reaction if the initial pressure of CO2 gas was 0.796 atm and the final pressure of CO2 gas is 0.394 atm. moles of CO2: molWhat is the mass percentage of CaO in the solid mixture? mass percent CaO: % What is the mass percentage of BaO in the solid mixture?mass percent BaO: %arrow_forwardConsider the following balanced reaction: 2 Z(g) + 4 A(g) → 3 Y(g) + 2 B(g) Occurs at STP a. 4.65 mol of Gas Z reacts with excess Gas A, what volume Gas Y will be made in L? b. If 5.76 mol of Gas Z reacts with 10.95 mol Gas A, identify the limiting reactant and calculate the volume of Gas B made in L.arrow_forward
- 8barrow_forward5. You are given 1.56 g of a mixture of KClO3 and KC1. When the mixture is heated the oxygen gas liberated displaces 327 mL of water at a temperature of 19°C. The total pressure of the gas in the collection flask is 735 mmHg. What is the percentage of KClO3 in the mixture?arrow_forwardA sample of CO2 occupies a volume of 14.87 liters at 140.5 kPa. What pressure in kPa would the gas exert if the volume was decreased to 2.47 L?arrow_forward
- The flame produced by the burner of a gas (propane) grill is a pale blue color when enough air mixes with the propane (C3H8) to burn it completely. For every gram of propane that flows through the burner, what volume of air is needed to burn it completely? Assume that the temperature of the burner is 195.0°C, the pressure is 1.20 atm, and the mole fraction of O2 in air is 0.210. ____ Larrow_forwardThe Haber Process synthesizes ammonia at elevated temperatures and pressures. Suppose you combine 1580 L of nitrogen gas and 4590 L of hydrogen gas at STP, heat the mixture to run the reaction, then isolate the ammonia from the reaction mixture. What volume of NH₃ , measured at STP, would be produced? Assume the reaction goes to completion. N₂ (g) + 3 H₂ (g) → 2 NH₃ (g)arrow_forward0.17 moles of hydrogen gas and 0.33 moles of nitrogen gas are created from thereaction 2NH3 (g) → 3H2 (g) + N2 (g). The final total pressure in the reaction vessel is0.75 atm. Calculate the partial pressure of the hydrogen gas at the end of the reaction.Assume the number of moles of NH3 (g), left after the reaction occurs, is negligible.arrow_forward
- Pparrow_forwardGaseous ammonia is synthesized from nitrogen and hydrogen by the reaction N2(g) + 3H2(g)→ 2NH3(g) Assume you take 355 L of H2 gas at 25.0 °C and 542 mm Hg and combine it with excess N2 gas. a )What is the theoretical yield of NH3 gas? b) If this amount of NH3 gas occupies a 125-L tank at 25.0 °C, what is its pressure?arrow_forward4. In a laparoscopic surgery, carbon dioxide gas is used to inflate the abdomen to separate the internal organs and the abdominal wall. If the CO2 injected into the abdomen produces a pressure of 17 mmHg and a volume of 2.9 L at 26°C, how many moles of CO2 were used? How many grams? 5. What is the total pressure, in millimeters of mercury, of a gas mixture containing krypton gas at 0.45 atm and neon gas at 650 mmHg?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY