
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Can you please answer sub questions A and B and show all of the steps please and thank you
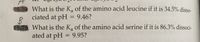
Transcribed Image Text:What is the K, of the amino acid leucine if it is 34.5% disso-
ciated at pH = 9.46?
%3D
What is the K, of the amino acid serine if it is 86.3% dissoci-
ated at pH = 9.95?
%3D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 5 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Think about the situation where you are investigating the relationship of study time vs. exam grade.a. Which variable would be on the x-axis (and why)?b. Which variable would be on the y-axis (and why)?c. Would you expect the slope to be positive or negative (and Why)? 2) Contrast and compare interpolation with extrapolation. Which is “safer” to use and why?3) Look up how “the best fit straight line” is constructed. The term “Least squares” should be in your explanation.4) If I plot some data and end up with a r2 of (-) 0.99, is my plot useless? Explain.arrow_forwardshow-all-working-explaining-detailly-each-step Answer should be typewritten using a computer keyboard.arrow_forward2:59 AT&T Aa 2:58 PM, Apr 22 Save Share II H3G H3C H30* ? Brz H3C SCH3arrow_forward
- I know you don't answer graded questions and I am not looking for you to straight up give me the answer to this but how do I even start? like I said before I know that the highest peak is 16amu which is Oxygen but do I just the vertical bar as a ratio? Like the next bar to the left (15amu?) is like 90% intensity? so like 9:10 ratio? There isn't even a 15amu element so please just give me some idea where to start. The place in the book it tells you to go to read has nothing about this stuff, its about finding empirical formulas from mass or percentages.... please help me!arrow_forwardAll changes save 7. When two solutions are mixed, a color change occurs. The data tables show the time between mixing and the color change for two sets of conditions. For Condition One, the solution concentrations were constant and temperature varied. For Condition Two, the temperature was constant and concentrations varied. Condition One: Concentration Time for Temperature (°C) Sample Color to Change 1 10° 36 sec 22° 14 sec Condition Two: Temperature Time for Concentration Sample Color to % Change 1. 100% 15 sec 2. 50% 24 sec Which of these statements is true according to the data? O The reaction rate is greater at 22°C than at 10°C. O Reducing the temperature increases the rate of the reaction. The reaction is affected by changes in temperature not by changes in concentration. O Decreasing the concentration increases the reaction rate. PREVIOUS 17 of 25 NEXT SAVE & EXITarrow_forwardIn the glucose molecule, why is the oxygen at carbon 5 the one that reacts with carbon 1 when there is an additional carbon at the end of the chain, carbon 6? t Predictions: On a. Due to the chirality of the last carbon, carbon 6 b. Because carbon 5 is the last chiral carbon 4 C. Because the oxygen at carbon 5 is in alpha position just like the oxygen at carbon 1 Accessibility: Good to go O 8: 14 101 f5 % 5 f6 A 17 40 & 7 18 Ö 7 W f9 8 8 f10 9 19 11 F ) O n f12 81°F Claarrow_forward
- In the last step, why is there a 2 in the second bracket before you input the numbers?arrow_forward7. Don believes that fewer than one-third of the students at his college hold part-time jobs while in school. To investigate this, he randomly surveyed 116 students and found that 32 students held part-time jobs. a. ( - Provide a 90% confidence interval for the population proportion of students that hold a part-time job. b. 1 (a). ;Give the correct interpretation of the interval in part c. Extra Credit: Does this interval support Don's claim? Why?arrow_forwardMust answer all questions eslse downvote A. MULTIPLE CHOICE. Choose the BEST answer. PLEASE HELP ME ANSWER EVERYTHING THANK YOU Q)What is used for heating small amounts of solids at a high temperature?a) Mortar and pestleb) Evaporating dishc) Crucible and coverd) Clay triangleQ)Which of the following is used in separation techniques?a-Rubber policemanb-Graduated cylinderc-Volumetric flaskd-Filter paperQ)Which of the following describes a centrifugate?a-Always clearb-Supernatant liquidc-Discarded via decantation onlyd-Solid particlesQThe inward force that pulls substances towards its center is called .a-Gravitational forceb-Centrifotal shiftc-Continental shiftd-Centrifugal forceQA "slippery floor" is considered a .a-hazardb-riskc-flash pointd-toxicantQ)Mrs. Lily Potter is 24 weeks pregnant. She was exposed to a chemical while making a potion. What is the type of the chemical she should be avoiding?a-neurotoxicantb-asphyxiantc-teratogend-carcinogenQ)Which of the following should be…arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY