
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
#23.
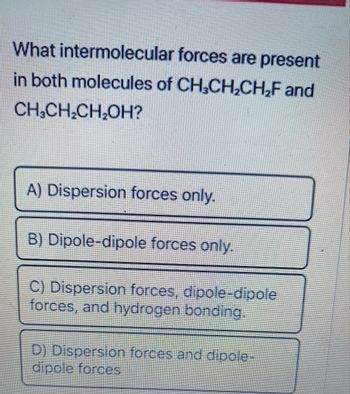
Transcribed Image Text:What intermolecular forces are present
in both molecules of CH₂CH₂CH₂F and
CH₂CH₂CH₂OH?
A) Dispersion forces only.
B) Dipole-dipole forces only.
C) Dispersion forces, dipole-dipole
forces, and hydrogen bonding.
D) Dispersion forces and dipole-
dipole forces
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- e OL 40% O A moodle.lsua.edu Alanie Fontenot JA What is the functional group found in the following molecule? N(CH3)2 Select one: O a. amine O b. amino acid O c. ketone O d. amide O e. ether Next page earrow_forward26. Benzene is an example of a(n), compound. O a. aliphatic b. aromatic c. nonaromatic d. more than one choice is correctarrow_forwardFor the given molecule, an amino alcohol, how many sigma bonds are present? Select one: O a. six O b. ten O. thirteen O d. sixteen O e. nineteenarrow_forward
- 26.arrow_forward*N(CH3)2 This molecule, which is used in the manufacture of medications, would be best-named: Select one: a. Dimethylphenylamine O b. Dimethylbenzamide O C. Benzimine Od. Dimethylbenzene Oe. Methylbenzanone Clear my choicearrow_forward36. Which of these formulas is(are) not valid for hexane? A. CH14 B. C6H6 C. C6H16 D. CH3CH2CH2CH2CH2CH3 E. CH3(CH2).CH3arrow_forward
- Please don't provide handwriting solutionarrow_forwardThe following essential oil is steam-distilled from heartwood chips, and its perfume is a pleasant blend of wood and flower with spicy undertones: a. rosewood b. rosemary c. pine d. cypressarrow_forward- Name the following structures: a. b. CN OH OH C1arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY