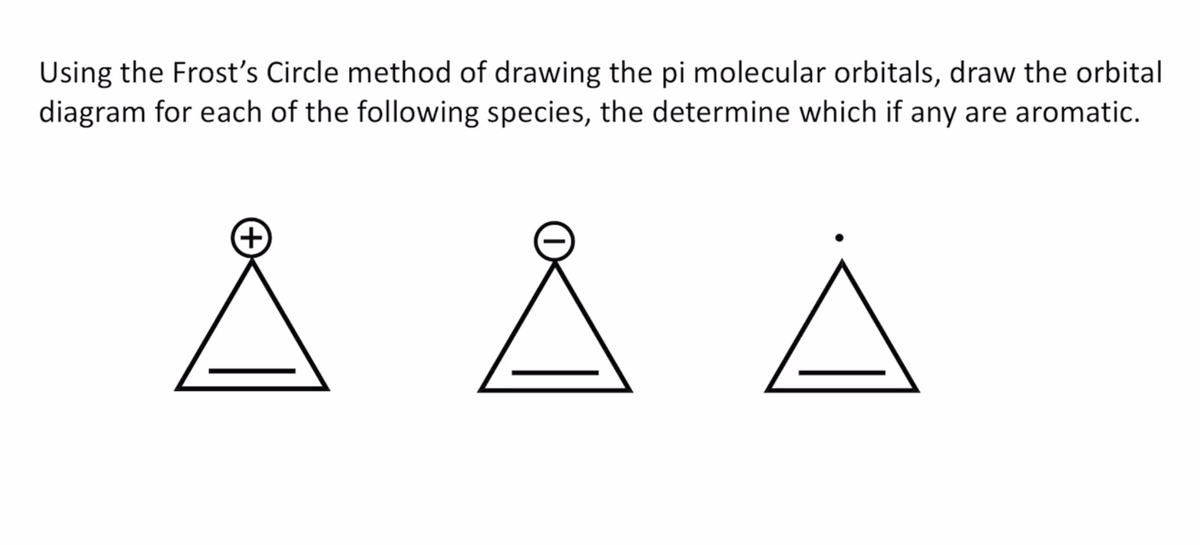
Using the Frost's Circle method of drawing the pi molecular orbitals, draw the orbital diagram for each of the following species, the determine which if any are aromatic. Å Å Å
Q: "Synthesis gas" is a mixture of carbon monoxide and water vapor. At high temperature synthesis gas…
A: Given,
Q: Which of the following aqueous solutions are good buffer systems? (Select all that apply.) 0.12 M…
A: A buffer is a solution which resists any change in pH on adding a small amount of acid or base.It is…
Q: Predict the major products formed when the compound shown below undergoes a reaction with 1…
A: Alkenes on treatment with HBr gives bromoalkanes and the addition occurs according to Markovnikov's…
Q: HO"NCI 2) H₂ C+ NaOH O-H
A:
Q: draw all possible structures for electrons in 3d orbitals
A: for d-orbitals, l=2Hence, m=-2,-1,0,1,2on the basis of probability calculations there are 5 possible…
Q: plete the following dislocation reactions: a/2[111] + a/2[1-1-1] --> a/6[11-1] + a/3[112] -->…
A: Given the dislocation reactions:a/2[111] + a/2[1-1-1] -->>a/6[11-1] + a/3[112]…
Q: Amines are weak bases. Table 10.2 in the course textbook provides a list of pKb values for several…
A: The objective of the question is to understand why increased substitution on nitrogen results in an…
Q: Sodium fluoride can be used to prevent dental cavities. What is the pH of 0.65 M NaF? Be sure your…
A: Given that,Sodium fluoride (NaF) can be used to prevent dental cavities. Sodium fluoride is a salt…
Q: In the movie, the white and green bonds come together and pull apart, and they just move around the…
A: No chemical reaction taking place Explanation:If they are simply coming together and moving, there…
Q: O OH 0 OEt Heat (-OH₂) OEt NaOCH2CH3 CH3CH₂OH NaOBut CH₂NO₂
A: The first reaction is an elimination reactionThe second reaction is intramolecular condensation…
Q: reasonable reaction sequence to use to synthesize it.
A: Given is organic synthesis reaction. The given starting compounds have functional groups like…
Q: 1.03 2. DMS
A: Ozonolysis is a chemical reaction that involves the cleavage of carbon-carbon double or triple bonds…
Q: A natural water sample contains 0.484 M NaCl, 054 M MgBr2 and trace levels Ag+ ions. Write the…
A: Ionic strength of the water sample = 2.1 MExplanation:As Ag+ ion is present in very less amount so…
Q: What size combinatorial library could be constructed using the substituents and diversity sites in…
A: The objective of this question is to identify substituents and diversity sites and calculate the…
Q: Black Forest Biologicals, a biotech startup, has a promising Alzheimer's drug candidate Compound…
A:
Q: Write the systematic (IUPAC) names for the amines. The names should have the format alkanamine.…
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: OMS CH2CH2OH ?
A: Substitution reactions are of two types i.e. SN1 and SN2.In SN1 formation of carbocation takes place…
Q: Consider the following reactants: X oyes X 5 Would elimination take place at a significant rate…
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: Draw the entire extended reaction mechanism (curly arrows) for this reaction но OH HO HO H OMe OMe…
A: The given reaction explains the acid catalysed protection of alcohol. The detailed mechanism is…
Q: 1. For each molecular geometry, give the number of total electron pairs, the number of bonding pairs…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Draw the major product of the following reaction. Br2 hv
A: Answer:Free radical substitution reaction takes place in presence of light and formation of free…
Q: Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and charg
A: The conversion of carbonyl compounds into alkene by the reaction with the triphenyl phosphonium…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting structure,…
A: In an organic reaction mechanism, a curved arrow represents the transfer of electrons. In an organic…
Q: For each of the substrates below, identify whether: (A) the rate of substitution doesn't depend on…
A: For substrate first, we can observe that .substitution will occur via SN1 reaction mechanism.
Q: Content/24SSEM_CHEM_114 X Aktiv Chemistry - Work b Home | bartleby x + ← → с app.aktiv.com Question…
A: The objective of the question is to determine which electron transition in a hydrogen atom would…
Q: There are TWO reactions that will happen when we react the molecule shown below with methanol under…
A: Trans-esterification:When ester in treated with excess alcohol in presence of acid catalyst, a new…
Q: The preparation of an aqueous solution is described in the table below. For this solution, write the…
A: Given,0.14 mol NaOH is added to 1.0 L of a solution that is 1.3 M in both NH3 and NH4Br. acids:…
Q: If the rate constant for production of NO gas and Cl2 gas from NOCl is 9.3 x 10 -6/sat 350 k and…
A: The reaction taking place is given as,=> 2 NOCl (g) ------> 2 NO (g) + Cl2 (g)Given : Rate…
Q: Propose a mechanism to account for the following reaction of an enamine with an alkyl halide: N-…
A: The reactivity of enamines as nucleophiles is due to the dipolar character of the one of the…
Q: - Which of the following is a meso compound? a. CI OH b. CH₂CH₁₂ OH CI CH.CH C. d. H₂C CH3
A: Meso compounds are the achiral compounds having multiple chiral centers. Meso compounds are…
Q: Draw the starting reactant that would produce this alcohol when treated with this reagent. Ignore…
A: The objective of the question is to find the starting reactant molecule of the reaction by which…
Q: 1. NaOCH3, CH₂OH, heat 2.. H3O+ quench
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 12.20 Propose an efficient synthesis for each of the following transformations HO (a) JOH (b) Br…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: ← Problem 22 of 30 Submit Draw the major product of this reaction. Ignore inorganic byproducts. H 1.…
A: Tollens’ reagent is a colorless, basic, aqueous solution containing silver ions coordinated to…
Q: Why is water avoided in the Grignard Synthesis of Benzoic Acid experiment? Write an equation for the…
A: Grignard reagent is an organometallic compound formed by the reaction of alkyl/aryl halides with…
Q: All the following reactions follow the law of conservation of mass except--- 4CO2 + 2H2O 2Fe2O3…
A: Find out the reaction those who do not follow the law of conservation of mass
Q: Consider the following reaction between calcium oxide and carbon dioxide: CaO (s) + CO2 (g)…
A: The objective of this question is to determine the theoretical yield of the reaction between calcium…
Q: 1. (a) Compound A,B and C are isomers with molecular formula of C4H8O. When compound A,B and C…
A: The objective of the question is to deduce the structures of compounds A, B, and C based on their…
Q: 2. If the compound of interest had to be distilled under reduced pressure, what modification could…
A: Distillation is a fundamental technique in chemistry used for separating and purifying substances…
Q: -NH Br + OH Draw the molecules on the canvas by choosing buttons from the Tools (for bonds and…
A: Final product given below Explanation:Step 1:- .
Q: 4) Which are expected to be the major product(s)? Draw the reaction mechanism. HBr
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: Write the acidic equilibrium equation for HClO. Be sure to include the proper phases for all species…
A: The objective of the question is to write the acidic equilibrium equation for HClO, including the…
Q: Predict the major product of the following reaction. A B 11 IV OH H₂O H₂SO4 OH ווו E OH IV Give…
A: This is hydration reaction. Alkene react with H+ and H2O to give water. Addition of water take place…
Q: 17. Arrange the compounds in each series in order of increasing acid strength: (a) AsH3, HBr, KH,…
A: Note: Since you have posted multiple questions, we will provide the solution only to the first…
Q: is the formula of h20
A: To find the formula for h20
Q: 3. To draw relevant arrow-pushed stepwise mechanism: OH Br Il e Me₂S -CH2 ཚིག · → ༢, Ph + Ph
A: Corey-Chaykovsky reactionIntramolecular nucleophilic substitution reaction
Q: Acyl transfer (nucleophilic substitution at carbonyl) reactions proceed in two stages via a…
A: Under acidic condition the carbonyl group gets activated by protonation followed by attack by the…
Q: A natural water sample contains 0.484 M NaCl, 054 M MgBr2 and trace levels Ag+ ions. Write the…
A: I have given answers and explanations for the questions that you have asked with my knowledge and…
Q: rand name of soft drink oncentration of undiluted standard phosphate solution, M 3x/03 M Solution…
A: The objective of this question is to calculate the concentration of phosphoric acid in the undiluted…
Q: A natural water sample contains 0.484 M NaCl, 054 M MgBr2 and trace levels Ag+ ions. Write the…
A: a) NaCl -> Na+ + Cl-MgBr2 -> Mg2+ + 2Br-Ionic strength (m) of the water sample can be…
Step by step
Solved in 3 steps with 3 images

- Compare the bonding in formic acid (HCOOH) with that in its conjugate base formate ion (HCOO). Each molecule has a central carbon atom bonded to the two oxygen atoms and to a hydrogen atom. Draw Lewis diagrams, determine the steric numbers and hybridization of the central carbon atom, and give the molecular geometries. How do the orbitals differ in formic acid and the formate molecular ion? The bond lengths of the CO bonds in HCOOH are 1.23 (for the bond to the lone oxygen) and 1.36 (for the bond to the oxygen with a hydrogen atom attached). In what range of lengths do you predict the CO bond length in the formate ion to lie?1/ Show how the participating p orbitals interact to form the highest energy pi molecular orbital of benzene. 2/ Use the polygon rule to draw the MO energy diagram of the cyclononatetraenyl anion. Assuming planarity, would this ion be aromatic or antiaromatic?The molecule shown on the right in the example in the right column is the amino acid histidine, and the five-membered ring is known as aromatic. An aromatic ring has 2, 6, 10, 14, etc., electrons placed in 2p orbitals around a ring. Indicate which of the following statements must therefore be true. 1. There are a total of six electrons in the pi system (defined as electrons in 2p orbitals), including the lone pair on the ring N that is not circled. 2. There are a total of six electrons in the pi system, including the lone pair on the ring N atom that is circled. 3. The lone pair on the ring N atom that is not circled resides in an sp2 orbital on an sp2 hybridized nitrogen atom. 4. Statements 2 and 3 are both true
- Which of the following represents the lowest energy pi molecular orbital for cyclopentadiene?Answer the following questions for 1,3,5- hexatriene, the conjugated triene containing six carbons. Which MOs are the frontier molecular orbitals?Answer the following question for 1,3,5- hexatriene, the conjugated triene containing six carbons. Which MOs are the frontier molecular orbitals?
- Which of the following criteria satisfies non-aromaticity? Choose whichever is applicable. (can be more than one answer) A. Continuous conjugation B. Noncontinuous conjugation C. Pi electrons are equal to 4n+2 D. Pi electrons are equal to 4n E. Planar geometryExplain how to construct the molecular orbitals of a conjugated cyclic system similar tobenzene and cyclobutadiene. Use the polygon rule to draw the energy diagram, and fillin the electrons to show whether a given compound or ion is aromatic or antiaromatic.Aromatic compounds must have a p orbital on every atom in the ring. Define this ?
- For the aromatic compounds below, draw the p-orbitals for the compound.Propose structures for molecules that meet the following descriptions:a. Contains two sp2-hybridized carbons and two sp3-hybridized carbons.b. Contains only four carbons, all of which are sp2-hybridized.c. Contains two sp-hybridized carbons and two sp2-hybridized carbons.Isn't the carbon where the charge changes sp3 hybridized? Wouldn't that make them all non-aromatic? Also, do we need to specify E and Z for the double bonds. Thanks.



