
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
thumb_up100%
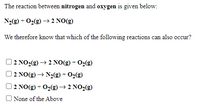
Transcribed Image Text:The reaction between nitrogen and oxygen is given below:
N2(g) + O2(g) → 2 NO(g)
We therefore know that which of the following reactions can also occur?
| 2 NO2(g) → 2 NO(g) + O2(g)
2 NO(g) → N2(g) + O2(g)
2 NO(g) + O2(g)→ 2 NO2(g)
O None of the Above
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In a coffee-cup calorimeter, 110.0 mL of 1.2 M NaOH and 110.0 mL of 1.2 M HCl are mixed. Both solutions were originally at 22.5°C. After the reaction, the final temperature is 30.5°C. Assuming that all the solutions have a density of 1.0 g/cm³ and a specific heat capacity of 4.18 J/°C.g, calculate the enthalpy change for the neutralization of HCl by NaOH. Assume that no heat is lost to the surroundings or to the calorimeter. AH = 0.182 kJ/molarrow_forwardSuppose a pair of chemical compounds A and B can react in two different ways: A+B C Reaction 1 gives product C. A+B D Reaction 2 gives product D. The following facts are known about the two reactions: . Both reactions are exothermic, but the heat of reaction of Reaction #1 is larger. • If a reaction vessel is charged ("filled") with A and B, then at first C is produced faster than D. Use these facts to sketch a qualitative reaction energy diagram for both reactions. Note: because these sketches are only qualitative, the energies don't have to be exact. They only have to have the right relationship to each other. For example, if one energy is less than another, that fact should be clear in your sketch. energy ^ A + B Reaction 1 reaction coordinate energy ^ A+B Reaction 2 reaction coordinate X Śarrow_forwardDetermine the energy associated with each of these processes in order to recycle 1.26 mol Cu. The smelting of copper occurs by the balanced chemical equation: CuO(s)+CO(g)Cu(s)+CO2(g)where ΔH°f,CuO is = –155 kJ/mol. Assume the process of recycling copper is simplified to just the melting of the solid Cu starting at 25°C. The melting point of Cu is 1084.5°C with ΔH°fus = 13.0 kJ/mol and a molar heat capacity, cP,Cu = 24.5 J/mol·°C.arrow_forward
- The aluminum cup inside your calorimeter weighs 41.55 g. You add 59.21 g of 1.0 M acetic acid solution and 50.03 g of 1.0 M sodium hydroxide solution to the calorimeter. Both solutions have an initial temperature of 19.9 oC, and the final temperature after addition is 26.8 oC. What is the molar enthalpy of neutralization, in units of kJ/mol? Assume that: the calorimeter is completely insulated the heat capacity of the empty calorimeter is the heat capacity of the aluminum cup: 0.903 J g-1 oC-1. the density of the two solutions is the same as that of water: 1.00 g/mL. the heat capacity of the two solutions is the same as that of water: 4.184 J g-1 oC-1. Perform all calculations without rounding, but then provide your answer to the correct number of significant figures.arrow_forwardThe copper mineral chalcocite, Cu2S, can be converted to copper simply by heating in air: Cu,S(s) + 0,(g) → 2Cu(s) + SO, (g)arrow_forwardA student mixes 67.0 mL of a 2.01 M sodium hydroxide solution with 22.4 mL of 6.45 M hydrochloric acid. The temperature of the mixture rises 17.2 ° C. The density of the resulting solution is 1.00 g mL and has a specific heat capacity of 4.184 J g · ° C . The heat capacity of the calorimeter is 16.97 J ° C . Part 1: (a) Identify the limiting reagent for the reaction. Part 2: (b) Calculate the heat of reaction (in J). qrxn = × 10 JEnter your answer in scientific notation. Part 3 out of 3 (c) Find the enthalpy of neutralization (in kJ/mol). ΔHneutralization = ____ kj/molarrow_forward
- Measurements show that the enthalpy of a mixture of gaseous reactants increases by 214. kJ during a certain chemical reaction, which is carried out at a constant pressure. Furthermore, by carefully monitoring the volume change it is determined that 62. kJ of work is done on the mixture during the reaction. Calculate the change in energy of the gas mixture during the reaction. Round your answer to 3 significant digits. Is the reaction exothermic or endothermic? 0 kJ exothermic endothermic x10 X Ś U!! 00arrow_forwardThe reaction between ammonia and oxygen is given below: 2 NH3(g) +2 O2(g) → N2O(g) +3 H₂O(1) We therefore know that which of the following reactions can also occur? N2O(g) +3 H2O(l) → 2 NH3(g) +2 O2(g) 4 NH3(g) +5 O2(g) →4 NO(g) + 6 H₂O(g) 4 NO(g) +6 H2O(g) →4 NH3(g) +5 O2(g) None of the Abovearrow_forwardThere are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. In the first step, calcium carbide (CaC₂) and water react t form acetylene (C₂H₂) and calcium hydroxide: + Ca(OH)₂ (s) CaC₂ (s) + 2H₂O(g) → C₂H₂(g) 1 In the second step, acetylene (C₂H₂), carbon dioxide and water react to form acrylic acid (CH₂CHCO₂H): 6C₂H₂(g) + 3C0₂(g) + 4H₂O(g) - 5 CH₂CHCO₂H (g) 1 Suppose the yield of the first step is 88.% and the yield of the second step is 86.%. Calculate the mass of calcium carbide required to make 9.0 kg of acrylic acid. Be sure your answer has a unit symbol, if needed, and is rounded to 2 significant digits. 0 Explanation Check ☐ x10 00 ロ・ロ © 2023 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Accearrow_forward
- 1) When HCl(aq) and NaOH(aq) are mixed in a beaker, the beaker feels warm to the touch. What is known about the enthalpy of this reaction? The reaction is exothermic. ΔH is positive. The reaction is endothermic. Heat is absorbed from the surroundings. 2) Consider this combination reaction:2Mg(s)+O2(g)→2MgO(s) ΔH=−1204 kJ What is the enthalpy for the decomposition of 1 mole of MgO(s) into Mg(s) and O2(g)? -602 kJ/mol 602 kJ/mol -1204 kJ/mol 1204 kJ/mol 3) The enthalpy for the formation of 1 mole of NH3(aq) is -80.29 kJ/mol. What is the enthapy for the formation of 3 moles of NH3(aq)? -240.87 kJ −518×103 kJ -26.76 kJ -83.29 kJarrow_forwardI know you this question has two parts but it counts as one question. Thank you so much and I hope you have a nice day.arrow_forwardCould someone please help! No plagiarism Please! 3. Draw an or find an image of an endothermic reaction or an exothermic reaction. Explain what makes it the type of reaction you chose to illustrate. Include arrows to show what direction the overall enthalpy of reaction (energy of reaction) flows. 4. Ammonium nitrate dissolves in water via the following reaction: NH4NO3(s) → NH4+(aq) + NO3-(aq) The bond energies of the compounds in the reaction are as follows: NH4NO3 = 3040 kJ/mol NH4+ = 1564 kJ/mol NO3- = 1009 kJ/mol How much total energy does the reaction have, and how will the environment change when the reaction occurs? Please show all of your workarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY