
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
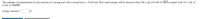
Transcribed Image Text:The enthalpy of neutralization for the reaction of a strong acid with a strong base is –56 kJ/mol. How much energy will be released when 306.1 mL of 0.400 M HCl is mixed with 143.1 mL of
0.500 M NaOH?
Energy released =
kJ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. Is heat considered a product or a reactant of the reaction?2. Is the formation of FeSCN+2 an endothermic or exothermic reaction?arrow_forwardAn ideal gas in a cylinder fitted with a piston expands at constant temperature from a pressure of 9 atm and a volume of 4.,0 L to a final volume of 13 L against a constant opposing pressure of 1.4 atm. How much heat does the gas absorb, expressed in units of L atm (liter x atm)? Latmarrow_forwardIn the following reaction, how much heat is generated when 3.21 moles of CH4 are burned? CH4 (g) + 2 O2 (g) → CO2 (g) + 2 H2O(g) AH° = -802 kJ/molarrow_forward
- Consider an endothermic reaction that takes place in 303 mL of solution. If the reaction requires 4,468 J of energy and the initial temperature of the solution is 26.7, what will the final solution temperature be? (3 sf) The density of solution is 1.00 g/mL and the specific heat of the solution is the same as that of water.arrow_forwardA 5.43 g sample of an unknown salt (MM = 116.82 g/mol) is dissolved in 150.00 g water in a coffee cup calorimeter. Before placing the sample in the water, the temperature of the salt and water is 23.72°C. After the salt has completely dissolved, the temperature of the solution is 28.54°C. If -3.13 x 10 J of heat was lost during the dissolution reaction of 0.0465 moles of the unknown salt, what is the enthalpy change (in kJ/mol of salt) for the dissolution reaction? Type here to searcharrow_forward15. The enthalpy change for the reaction CH,(g) + 20,(g) CO,(g) + 2H,O() is -891 kJ for the reaction as written. a. What quantity of heat is released for each mole of water formed? b. What quantity of heat is released for each mole of oxygen reacted?arrow_forward
- In order to break water into hydrogen and oxygen, water is heated to more than 500°C. Which kind of reaction is this and why? It is exothermic because heat needs to be released by the reactants to form the products. It is endothermic because heat needs to be released by the reactants to form the products It is exothermic because heat needs to be absorbed by the reactants to form the products. It is endothermic because heat needs to be absorbed by the reactants to form the products.arrow_forward3. A piece of metal weighing 59.047 g was heated to 100.0 °C and then put it into 100.0 mL of water (initially at 23.7 °C). The metal and water were allowed to come to an equilibrium temperature, determined to be 27.8 °C. Assuming no heat lost to the environment, calculate the specific heat of the metal.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY