
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
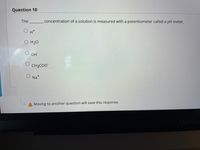
Transcribed Image Text:Question 10
The
concentration of a solution is measured with a potentiometer called a pH meter.
H*
O H20
OH
CH3CO"
Na+
A Moving to another question will save this response.
MacBoo
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Consider two aqueous solutions of equal concentration: chlorous acid (HCNO, K, = 2.0 x 10 4) and acetic acid (CH3COOH, K, = 1.77 x 10-5). Which statement is true when the solutions are at equilibrium? %3! O CNO produces more [OH ] than CH3COO O HCNO produces less [H3O*] than CH3COOH O The pH value of HCNO solution is the same as that of CH3COOH solution. O HCNO produces more [H3O*] than CH;COOH O The pH value of HCNO solution is higher than that of CH3COOH solution.arrow_forwardThe OH concentration in an aqueous solution at 25 °C is 6.1 x 10-5. What is [H*]? Marrow_forward1.00 mL of 0.345 M HCI -Solution A. To this solution you add 123.00 mL of H2O -Solution B. i) Solution A, determine the hydronium (H^ + ) and hydroxide (OH^-) concentrations And the pH and pOH ii) Solution B determine the hydronium (H ^+ ) and hydroxide (OH) concentrations and the pH and pOHarrow_forward
- What are the equilibrium concentrations of NH3, NH4+, and OH in a 0.78 M solution of ammonia? Kb = = 1.8 × 10-5 [OH-] [NH₂+] = [NH3] = What is the pH of the solution? pH = M M Marrow_forward||| ctrl lacc shift ↑ tab caps lock VILION Mc ALE Graw HI esc K →1 Pavilion x360 fn O CHEMICAL REACTIONS Determining the molar mass of an acid by titration McCA X Eplanation g mol V An analytical chemist weighs out 0.188 g of an unknown triprotic acid into a 250 mL volumetric flask and dilutes to the mark with distilled water. She then titrates this solution with 0.1500M NaOH solution. When the titration reaches the equivalence point, the chemist finds she has added 38.4 mL of NaOH solution. Calculate the molar mass of the unknown acid. Be sure your answer has the correct number of significant digits. f1 Type here to search A https://www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IvUrTNdLZh5A8CnG03PBGuXr8iCPa7ZMmym f2 @ Z Check 2 L M W hp S ALE MCCA ALER f3 # Mc Graw 3 X alt D 4 $ 4 с x10 X R O f5 Me Graw MI % F 5 at 40 MCCA ALE ALE ● V T S f6 4- G 6 ■ T f7 B Y ♫+ & Mc 7 H raw fg MCCA ALER *. N KAA © 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center |…arrow_forwardConsider the “window cleaner” problem, question 2 on page 12 of your lecture notes. Suppose that prior to the titration, the 10 mL sample of window cleaner was diluted by adding about 10 mL of DI water so that the solution could have better contact with the pH meter. What effect would this dilution have on the volume of titrant needed? (a) The amount of titrant would be the same because the moles of analyte was not changed.(b) The amount of titrant would be doubled because the analyte solution volume was doubled.(c) The amount of titrant would be halved because the was diluted by half.arrow_forward
- it pt Calculate the pH of a solution that has an ammonium chloride concentration of 0.042 M and an ammonia concentration of 0.047 M. pH = Ky=1.8 x 10-arrow_forwardCalculate the molarity of an NaOH solution from the following titration data. Be sure the answer has the correct amount of significant figures. The chemical equation for this titration is as follows: NaOH + KHP ⟶ NaKP + H2O NaOH buret reading, inital: 15.27 mL NaOH buret reading, final: 8.32 mL Mass of KHP (204.22 g/mol): 1.1592 g Calculate the molarity of an NaOH solution from the following titration data. Be sure the answer has the correct amount of significant figures. The chemical equation for this titration is as follows: NaOH + KHP ⟶ NaKP + H2O NaOH buret reading, inital: 15.27 mL NaOH buret reading, final: 8.32 mL Mass of KHP (204.22 g/mol): 1.1592 garrow_forwardthe solution has to be acidified before testing for Ag+ ions. To make sure that the solution is acidic, what should be done? Apply a drop of the solution to red litmus paper to see if it turns blue. Apply a drop of the solution to blue litmus paper to see if it turns red. Apply a drop of the solution to pH paper to see if the pH is above 7. Apply a drop of the solution to your tongue to see if it tastes sour.arrow_forward
- a Calculate the molar concentration of OH in a solution that contains 8.7×10-¹2 M H30+. (Enter your answer to two significant figures.) [OH-] = M bCalculate the molar concentration of OH in a solution that contains 3.5×10-3 M H30+. (Enter your answer to two significant figures.) [OH-] = Marrow_forwardWhat is the concentration of H* in 0.042 M HCIO4? [H*] = i What is the pH of the solution? pH = M What is the OH- concentration in the solution? For scientific notations, use the format 1.0E-10, for example. [OH-] = i M By how much does the pH change if the concentration of H* is doubled? iarrow_forwardPlease answer all parts.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY