
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Transcribed Image Text:11
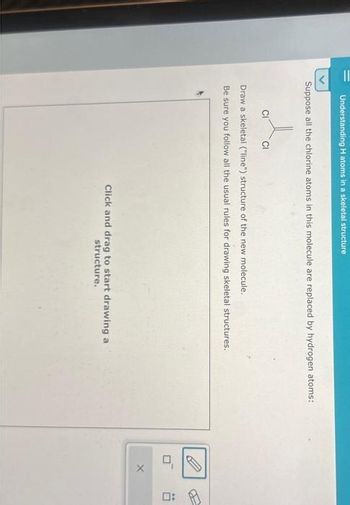
Understanding H atoms in a skeletal structure
Suppose all the chlorine atoms in this molecule are replaced by hydrogen atoms:
CI
Draw a skeletal ("line") structure of the new molecule.
Be sure you follow all the usual rules for drawing skeletal structures.
Click and drag to start drawing a
structure.
O
X
0: A
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Question 16.b of 25 Classify and describe the properties of the following nitrogen containing compound. Provide a systematic name for this structure. N,N,N- 2- N- eth tri hex bi di CH3 prop N.N- CH3 pent but meth al amide an 1-arrow_forwardWhat is a double bond? What is a triple bond? Compare the bond lengths for single, double and triple bonds between two carbon atoms.arrow_forwardSide by side dot structures of two water moleculs are given below. Draw structures like that on your paper. Put partial positive and partial negative charges where appropriate. Show two hydrogen bonds by connecting the partial positive charge on one molecule to the partial negaive charge on the other molecule using a dotted line. Upload your answer here. H-O-H H-O-Harrow_forward
- Question 3. Select the correct molecular geometry for each of the following central atoms: NH3= [Select] PCI4+ = NOCI= [Select] HNO2 (central atom = N) = [Select] HNO2 (central atom = O) = [ Select] SO3 [ Select] SO₂ = CO₂ = [Select] = [ Select] [ Select] O (> COF2= [Select] () CH3OH (central atom = C) = [ Select] ✪ CH3OH (central atom = O) = [ Select] O ◊ û NH₂CH₂CO₂H (central atom = N) = [Select] NH₂CH₂CO₂H (central atom = CH2) = [Select] NH₂CH₂CO₂H (central atom = CO2) = [Select] ✪ ◊ ✪ ✪ ✪arrow_forwardPlease help answer thisarrow_forwardPlease help mearrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY