
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Transcribed Image Text:cula...
=
18.3 Gibbs Free E...
O Kinetics and Equilibrium
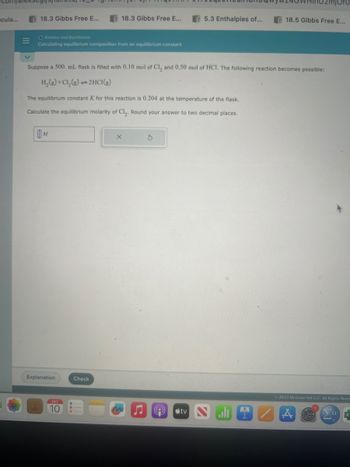
Calculating equilibrium composition from an equilibrium constant
Suppose a 500. mL flask is filled with 0.10 mol of Cl₂ and 0.50 mol of HCI. The following reaction becomes possible:
H₂(g) + Cl₂(g) -2HCl(g)
M
18.3 Gibbs Free E...
The equilibrium constant K for this reaction is 0.204 at the temperature of the flask.
Calculate the equilibrium molarity of Cl₂. Round your answer to two decimal places.
Explanation
DEC
10
Check
5.3 Enthalpies of... CP 18.5 Gibbs Free E...
X
tv Nall
© 2023 McGraw Hill LLC. All Rights Resea
A
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Suppose a 500. mL flask is filled with 1.5 mol of Cl2 and 0.60 mol of HCI. The following reaction becomes possible: H2(g)+ Cl2(g) 2HCl (g) The equilibrium constant K for this reaction is 3.73 at the temperature of the flask. Calculate the equilibrium molarity of H2. Round your answer to two decimal places. Омarrow_forwardSuppose a 250. mL flask is filled with 1.1 mol of CO, 0.80 mol of H,O and 0.60 mol of CO,. The following reaction becomes possible: CO(g) +H,0(g) - CO,(g)+H2(g) The equilibrium constant K for this reaction is 5.72 at the temperature of the flask. Calculate the equilibrium molarity of CO. Round your answer to two decimal places. | Marrow_forwardSuppose a 250. mL flask is filled with 0.50 mol of H, and 0.90 mol of HI. This reaction becomes possible: H, (g) +1,(g) = 2HI (g) Complete the table below, so that it lists the initial molarity of each compound, the change in molarity of each compound due to the reaction, and the equilibrium molarity of each compound after the reaction has come to equilibrium. Use x to stand for the unknown change in the molarity of H,. You can leave out the M symbol for molarity. H2 HI initial ? change equilibriumarrow_forward
- Suppose a 500. mL flask is filled with 1.7 mol of Cl₂, 1.1 mol of CHCl3 and 1.3 mol of HCl. The following reaction becomes possible: Cl₂(g) + CHCl3 (g) → HC1(g) +CC14 (g) The equilibrium constant K for this reaction is 9.41 at the temperature of the flask. Calculate the equilibrium molarity of Cl₂. Round your answer to two decimal places. 10999arrow_forwardSuppose a 250. mL flask is filled with 0.70 mol of Cl₂ and 1.1 mol of HCI. The following reaction becomes possible: H₂(g) + Cl₂(g) 2HCl(g) The equilibrium constant K for this reaction is 0.288 at the temperature of the flask. Calculate the equilibrium molarity of Cl₂. Round your answer to two decimal places. M 1arrow_forwardSuppose a 250. mL flask is filled with 1.5 mol of CO, 0.80 mol of NO and 0.70 mol of CO,. The following reaction becomes possible: 2' NO, (g) +CO(g) - NO(g)+CO,(g) The equilibrium constant K for this reaction is 6.92 at the temperature of the flask. Calculate the equilibrium molarity of NO. Round your answer to two decimal places. olo Ar OM ?arrow_forward
- Suppose a 500.mL flask is filled with 1.5mol of CO, 1.1mol of H2O and 1.6mol of H2. The following reaction becomes possible: CO(g)+H2O(g)+CO2(g)+H2(g) The equilibrium constant K for this reaction is 0.840 at the temperature of the flask. Calculate the equilibrium molarity of CO2. Round your answer to two decimal places.arrow_forwardSuppose a 250. mL flask is filled with 0.70 mol of H2 and 1.1 mol of 12. The following reaction becomes possible: H2(g) +I2(g) — 2HI(g) =0 The equilibrium constant K for this reaction is 8.37 at the temperature of the flask. Calculate the equilibrium molarity of 12. Round your answer to two decimal places. Омarrow_forwardSuppose a 250. mL flask is filled with 0.60 mol of SO₂ and 1.0 mol of SO3. This reaction becomes possible: 2SO₂(g) + O₂(g) — 2SO3(g) Complete the table below, so that it lists the initial molarity of each compound, the change in molarity of each compound due to the reaction, and the equilibrium molarity of each compound after the reaction has come to equilibrium. Use x to stand for the unknown change in the molarity of O₂. You can leave out the M symbol for molarity. initial change equilibrium SO₂ 0₂ 0 X 0 SO₂ 0 010 X Śarrow_forward
- Suppose a 500. mL flask is filled with 0.30 mol of SO, and 1.2 mol of SO3. This reaction becomes possible: 2SO, (g) +O,(g) 2SO3 Complete the table below, so that it lists the initial molarity of each compound, the change in molarity of each compound due to the reaction, and the equilibrium molarity of each compound after the reaction has come to equilibrium. Use x to stand for the unknown change in the molarity of O,. You can leave out the M symbol for molarity. so, SO2 O2 initial change equilibriumarrow_forwardSuppose a 500. mL flask is filled with 1.3 mol of Cl₂ and 0.80 mol of HC1. The following reaction becomes possible: H₂(g) + Cl₂(g) → 2HCl (g) The equilibrium constant K for this reaction is 9.14 at the temperature of the flask. Calculate the equilibrium molarity of H₂. Round your answer to two decimal places. M X Ś E A olo 18arrow_forwardSuppose a 500. mL flask is filled with 0.50 mol of N, and 0.90 mol of NO. This reaction becomes possible: N,(8) +O,(g) – 2NO(g) Complete the table below, so that it lists the initial molarity of each compound, the change in molarity of each compound due to the reaction, and the equilibrium molarity of each compound after the reaction has come to equilibrium. Use x to stand for the unknown change in the molarity of N,. You can leave out the Msymbol for molarity. NO initial change equilibriumarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY