
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Please don't provide handwrittin solution....
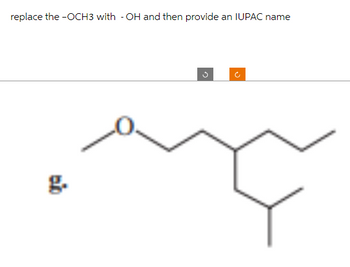
Transcribed Image Text:replace the -OCH3 with -OH and then provide an IUPAC name
ம்
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Sucrose is an example of weak-electrolyte. a) T b) F Select one: True O Falsearrow_forwardYou have to make 50 mL of 0.05M HCl from a 10M HCl stock solution, but the only measuring devices available are 100mL graduated cylinders and 10mL serological pipets. How could you accurately make the dilute HCl solution (clearly explain)?arrow_forwardWhat about any gasses that is created DURING the process of the reactants, and then lost/released while it is undergoing the reaction? Wouldn't that make all solutions suitable?arrow_forward
- 8. catalytic HCI water solventarrow_forwardraw Hill C C Chegg - Get 2 MOTION EYE OWLv2 | Assig Submit Answer Content gagenow.com/ilrn/takeAssignment/takeCovalentActivity.do?locator assignment-take OWLv2 | Orx Retry Entire Group [Review Topics] [References] Use the References to access important values if needed for this question. CengageNOW An aqueous solution is made by dissolving 17.2 grams of iron(II) chloride in 431 grams of water. The molality of iron(II) chloride in the solution is m. e 4 more group attempts remaining New Tabl D →1 D 2. Other Bocarrow_forwardExplain with detail some factors which must be considered when choosing a solvent to dissolve a solid powder.arrow_forward
- Pb(NO3)2 is placed in water. Please predict the physical state using our solubility rules from class. Select one: a. aqueous - (aq) b. liquid - (l) c. gas - (g) d. solid - (s) e. plasma - (p)arrow_forwardmixing instead of vigorously shaking them will help prevent emulsions from forming in the first place. 1. Questions 1. You are given a substance that is equally soluble in water and hexane (two immiscible solvents). a) What is K? b) If you start with 10 g of the substance dissolved in 100 mL of water and are given 100 mL of hexane to extract with, algebraically show that it is better to do two smaller extractions using 50 mL of hexane for each extraction than it is to do one extraction using 100 mL o hexane. 2. You have a solution that contains 4.0 g of oxalic acid dissolved in 100 mL of water. This solution is placed in a separatory funnel, and then 100 mL of diethyl ether is added to the separatory funnel and an extraction is performed. (Diethyl ether and water are immiscible solvents.) Use the following constants to calculate how much oxalic acid is in each solvent after the extraction. Solubility of oxalic acid in water = 9.5 g / 100 mL Solubility of oxalic acid in ether = 16.9 g /…arrow_forwardSelect one for each boxarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY