
Fundamentals Of Analytical Chemistry
9th Edition
ISBN: 9781285640686
Author: Skoog
Publisher: Cengage
expand_more
expand_more
format_list_bulleted
Question
Which compound is most consistent with the following mass spectrum? (choose the best from the figures below)
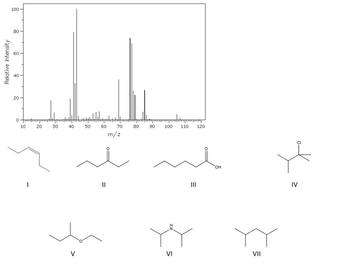
Transcribed Image Text:Relative Intensity
40
20-
100-
80
10
20
30
40
50
60
70
80
90
-2
m/z
VI
100
110
III
120
CH
VII
IV
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Similar questions
- Relative Intensity 100 80- 40 T 20- 0 MS-IW-0614 25 50 75 100 m/z 125 150 1४ og arbe Day OH OH 175arrow_forwardAssessment-Chem 18-Gen (X 80 $ 54 R UCI General Chemistry Peer Tu X 65.2% Use the molar bond enthalpy data in the table to estimate the value of AHin for the equation CCI, (g) + 2 F₂ (g) →→→ CF₂(g) + 2Cl₂(g) The bonding in the molecules is shown. ΔΗ; = 888 F4 ++++ % 5 FS F-F A 6 New Tab MacBook Air ܀ T Y F6 & 7 CICI CI-CI A4 F7 * 8 ∞ Resources Average molar bond enthalpics. (Hond) kJ. mol Bond 464 C=N 142 N-H 351 N-N 502 N=N 730 N=N 347 F-F 615 811 414 439 331 276 293 615 Bond O-H 0-0 C-0 0=0 C=0 C-C C=C C=C C-H C-F C-CI C-Br C-N C=N DII FB 69 DD F9 kJ Question Source: McQuarrie, Rock, And Gallogly 4e-General Chemistry Publisher: University Science Books 1 0 ) CI-CI Br-Br H-H H-F H-CI H-Br H-S S-S 0 A F10 I' Check Answer P kJ. mol-1 890 390 159 418 945 155 243 192 435 565 431 368 364 225 3 F11 0 + Show All 411) F12 X }arrow_forwardYou are trying to come up with a drug to inhibit the activity of an enzyme thought to have a role in liver disease. In the laboratory the enzyme was shown to have a Km of 1.0 x 10-6 M and Vmax of 0.1 micromoles/min.mg measured at room temperature. You developed an uncompetitive inhibitor. In the presence of 5.0 x 10-5 M inhibitor, the apparent Vmax was determined to be 0.02 micromoles/min.mg. What is the Ki of the inhibitor?arrow_forward
- a laccd sign i X C tab McGraw-Hil X A ALEKS-Shu X https://www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-lgNslkr7j8P3jH-IvUrTNdLZh5A8CnG03PBGuXr8iCPa7ZMmym9HtY-pEBmXkvCfq... O CHEMICAL REACTIONS Calculating ion molarity using solute mass shift ↑ Calculate the molarity of Cl anions in the chemist's solution. Be sure your answer is rounded to the correct number of significant digits. caps lock esc Explanation ALEKS A chemist prepares a solution of vanadium(III) chloride (VC13) by measuring out 1.50 g of VCl₂ into a 300. mL volumetric flask and filling to the mark with distilled water. ILION K →1 E mol L ? Type here to search A Graw X HI Check 12 (8) 2 Z W S # 3 X alt $ D 4 C x10 f5 % X 100 LO 5 V f6 4- (0) 6 ALEKS G 14.1 f7 4+ & Y B hp 7 X H McGraw-Hi x A ALEKS-Shix Sign In 시기 fg IAA * © 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibilit # a @ 76°F ㅅ 11:03 7/25/2 00 8 N 19 DII 9 fo K M DDI 2/5 0 P 12 insert [ = X + alt prt sc 46 Shusha 0 1 A backs paus ctearrow_forwardent arch X Aktiv Chemistry ADD FACTOR x( ) 4.82 × 104 10¹2 A laser pulse is 4.82 x 10-2 milliseconds How many nanoseconds is this? 10-⁰ 4.82 x 10-8 10⁰ ns 10 º 4.82 × 10² X Question 30 of 31 4.82 x 10-14 ART ms PeopleSoft session expired 10-12 4.82 x 10-5 10⁰ ANSWER 4.82 x 1016 4.82 x 10-² 4.82 x 101⁰ min 1015 X ? Anthropology and Archaeolo X μs 10³ 4.82 × 10-11 RESET 5 10-3 P 1arrow_forwardALEKS - Jacqueline Hoppenrey x G convert mg to g - Google Searc x A www-awn.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IQiHqRdYV 6Ux63Syp.JXz0Coxvwqgg4JkWI72X79QvOLp9_7U27sYQhkaocvdwecGvsUzo65uy3F6spORRg1XSqgh81is O STOICHIOMETRY Using molarity to find solute mass and solution volume Jacqueline A chemist adds 55.0 mL of a 4.75M silver perchlorate (AGCIO,) solution to a reaction flask. Calculate the mass in grams of silver perchlorate the chemist has added to the flask. Round your answer to 3 significant digits. Explanation Check Privacy Accessibil 2021 McGraw-Hill Education All Rights Reserved Terms of Usearrow_forward
- Relative Intensity 100 M+ = 132 80 60 40- 20- 0- 25 50 75 m/z 100 T 125arrow_forwardI 596420-7 x The impo X IG what ma X H PAP Cher X Qchapter x ぐ -> b ar-2903012.agilixbuzz.com/student/135113422/activity/9ff34d34-df47-4fas022 Mastery Assess It 7 PAP Chemistry-2903012-42100P-1/ Stoichiometry/Lesson 107 12. The combustion of glucose is represented by the following balanced equation: CGH12O6+6 0, 6 H20+6CO2. Which reactant is the limiting reagent if there is 1 gram of both 0, Hy06 and 0,7 O a. CgHy06 O b. H20 O c. 0, Od. CO,arrow_forwardI need step by step process solving [4.184x156(Tf-295)] + [4.184x85.2(Tf-368)] = 0. because my work is wrong. Thank you!!arrow_forward
- Subject- chemistryarrow_forwardA. Draw structure of compound. B. Label base peak and molecular ion peak.arrow_forward18P3JH-IQs_dp5pR4EN YC-70kXyvIz36BqJhw3sVPjBM1Gk45mlzq1R2r1PxBPnLctsC-Txx7dUax4sp-1P6W9HQV7MFFXM-1wpl2?1oBw7QYjlbavbSPXtx- Login | Student Veri. H Logout MyProgramming ab Imported From IE ITEC2110:Summer2... TunesToTube - Upl... Web Development.. O CHEMICAL REACTIONS Clap Using molarity to find solute mass and solution volume A chemist adds 0.35 L of a 0.21M potassium dichromate (K, Cr, O,) solution to a reaction flask. Calculate the mass in grams of potassium dichromate the chemist has added to the flask. Be sure your answer has the correct number of significant digits. IIarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning