
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Question 3
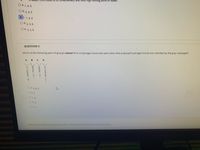
Transcribed Image Text:and thus hign bolling point of water.
O a. 1,4, 5
Ob. 2.3.5
OC 1,3, 6
O d. 2, 3, 6
O e. 3, 5, 6
QUESTION 3
Which of the following pairs of groups cannot form a hydrogen bond with each other (the proposed hydrogen bonds are indicated by the grey rectangle)?
A B CD
Oa. A & D
Ob.c
OCB
O e. A
Click Save and Submit to save and submít. Click Save All Answers to save all answers.
Com -0-
名 - O1
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Similar questions
- What is the scientific method answers to the freezing point of waterarrow_forwardConsider the following operations on the number 8.83 x 10-². Without using a calculator, decide which would give a significantly smaller value than 8.83 x 10-2, which would give a significantly larger value, or which would give essentially the same value. 8.83 x 10-2 +3.23 x 105 (same 8.83 x 10-2-3.23 x 105 smaller 8.83 x 10 2 x 3.23 x 105 larger v 8.83 x 10-2/3.23 x 105 smallerarrow_forwardLaboratory 2 Report Sheet-Lab 2 A.2 Area Your measurements Length = Width = Area = (Show calculations.) Why could two students obtain different values for the calculated areas of the same rectangle? A.3 Volume of a solid by direct measurement Shape of solid Formula for volume of solid 18 Height Width Another student's measurements Volume of the solid (Show calculations of volume including the units.) Length Diameter (if cylinder) Page 3 of 3 482 words 区arrow_forward
- Which statement is true? Question 12 options: Precision is measured by standard deviation. Precision is measured by error or relative error. Accuracy is measured by the standard deviation.arrow_forwardThat's not the correct answer check the math the answer is off by 100arrow_forwardFor the following reaction : H2 (g) + I2 (g) 2HI (g) it was found that a 13.0L vessel at equilibrium contained 0.852 mol H2, 0.361 mol I2, and 2.99 mol of HI. A) Write an expression for Kc. B). Determine the value of Kc for this reaction.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY