
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Kk.208.
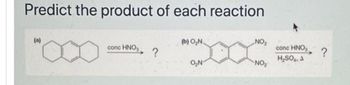
Transcribed Image Text:Predict the product of each reaction
(a)
conc HNO₂
?
(b) O₂N.
O₂N
NO₂
NO₂
conc HNO
H₂SO4. A
?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Kk.235.arrow_forwardDuring World War II, physicists developed methods to separate the uranium-235 and -238 isotopes. One method involved converting the uranium metal to a gas, UF6, and then allowing the gas to diffuse through a porous barrier, with the lighter gas diffusing faster. What is the difference in the speeds of the two UF6 gas species at room temperature?arrow_forwardChoose the correct answer. The difference between the massnumber of an isotope and its atomic number is (a) directlyrelated to the identity of the element; (b) the number of electrons;(c) the number of neutrons; (d) the number of isotopesarrow_forward
- 1. Write the Xnotation for each atomic depiction: (a) Z 18e 18p 20n% (b) 25e 25p* 30n (c) 47e 47p* 62narrow_forwardIs 1s22s22p6 the symbol for a macroscopic property or a microscopic property of an element? Explain your answer.arrow_forwardo Now.pdf Balance the following equations: (these chemical equations are unbalanced): 1) 2) 3) NaBr + H3PO4 PbSO4 → Mg + Open with Fe₂O3 Group 1 Na3PO4 + Group 2 PbSO3 + Group 3 Fe + HBr MgO ECarrow_forward
- Please explain how you can identify your target molecular compounds contain isotopes of chloride and bromine?arrow_forwardWhat is the relative abundance of isotope A?arrow_forward10 (c) An element T has a melting point of 30°C and a boiling point of 2440 °C. It conducts electricity at room temperature. It burns in oxygen to form an oxide with formula T₂O₂ which can react with both acids and bases. T also forms a compound with fluorine, which has a high melting point and conducts electricity in molten form. The approximate relative atomic mass of T is 70. (1) What type of oxide is T,O,? (i) Give two properties that indicate that T is probably a metal. 1. 2 (iii) Predict the formula for the fluoride of T E [1] [2] N [1] (iv) What are the products of the electrolysis of the molten fluoride of using inert electrodes? and (v) In which group and period of the Periodic Table will T be placed? group period 21 2) (vi) Write the symbol of the element in the Periodic Table which most closely resembles T.arrow_forward
- 6.) which of the following will have a product? Feo + Ni²* (aq) Au + Al³+ Al" → (aq) Nao + Zn?* + Zn²+ + (be) (aq) Al (s) 9 + Au" (aq)arrow_forwardAunt Julie went to the jewelry store to buy a gift for her amiga. As an aunt from Poveda, she knows that the karat scale is based on mass percentages. She was also informed by Uncle Bruno that pure gold is equivalent to 24 karats. If she bought a bracelet made of gold alloy that is 50 mol% silver and 50 mol% gold, what is the karat number of the bracelet?arrow_forwardThe decay series of Uranium 238 includes 3 isotopes. Which one has the greatest abundance in a sample of pitchblende ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY