
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
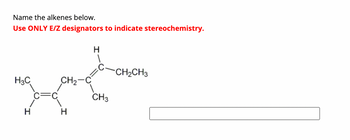
Transcribed Image Text:Name the alkenes below.
Use ONLY E/Z designators to indicate stereochemistry.
H3C
H
Ja
H
CH₂-C
H
CH3
CH₂CH3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- How many isomers (structural, geometric, and stereo) have the formula C5H10, and have "cyclo" in their name? (i.e. they contain a ring) 4 06 07 5arrow_forward#5arrow_forwardProvide the correct IUPAC name for the compound shown here. SEP 27 3- Delete H₂C. CH3 tv CH₂ CH3 -CH₂ CH3 4- 6- 1- 2- 5- di cyclo iso tri tert- sec- hept pent eth hex meth prop ene ane yne yl A 33159 ))) Warrow_forward
- Name the alkene below. Use ONLY E/Z designators to indicate stereochemistry. H H H3C CH3 CH;CH2CH2 CH3arrow_forwardIs the alkene in the molecule below CIS, TRANS, or neither? Cis Trans Neither CI HO CO₂Harrow_forwardA student drew the structural diagram shown below. CH, 2 HHHHHHHH 1 H-C-C-C-C-C-C-C-C-H III HHHHHHHH 3 CH₂CH₂CH₂ oct-3-ene Compounds that Contain Eight Carbons 2,3-dimethylhex-2-ene I 5 CH₂CH₂ 6 2,3-dimethyl-1-propylcyclopropane 7 CH₂ CH₂ 4 3,3-dimethylhexane The compounds above that are isomers of the structural diagram that the student drew are numbered ______ '__—_ › andarrow_forward
- Draw a structural formula for the alkene you would use to prepare the alcohol shown by hydroboration/oxidation. CH3 HO CH3 •You do not have to consider stereochemistry. •You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. H3C-CH3 n [ ?arrow_forwardWhich of the following is the lower energy cyclohexane conformer? A-values: Br = 0.6 kcal/mol; isopropyl = 2.1 kcal/mol. Œ A B Br B Brarrow_forwardSelect the most stable alkene that can be hydrogenated to form 1,2-dimethylcyclopentane: ㅂ 아 d 아 임 H2. Pd/Carrow_forward
- How do I name this compound? Specify (E)/(Z) stereochemistry, if relevant, for straight chain alkenes only.arrow_forwardsystematic (IUPAC) name: H3C-CH2-NH2-H3C-CH2-CH-CH2 I CH3 NH2 I CH-CH3 H3C-CH2-N-CH2-CH3 I H NH-CH3 I H3C-CH-CH3 H3C-N-CH3 I H2C-CH2 -CH-CH3 H3C-N-CH2-CH3-H3C-CH2-CH2-CH2arrow_forwardChem101 a app.101edu.co pen E SimpleBills ATu Howdy IT Canvas O Phleb course h Hulu all Gradescope 101 Chem101 Netflix Question 3 of 9 Provide the correct IUPAC name for the compound shown here. CH3 CH2=C-CH=CH2 2,3- 2- 1,3- 1,2- 3- di tert- tri iso cyclo sec- NOV Aa MacBook Pro G Search or type URL 6. 7 %3D T Y U * 00arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY