
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
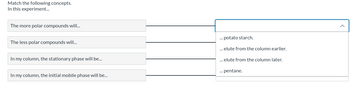
Transcribed Image Text:Match the following concepts.
In this experiment...
The more polar compounds will...
The less polar compounds will...
In my column, the stationary phase will be...
In my column, the initial mobile phase will be...
... potato starch.
... elute from the column earlier.
... elute from the column later.
... pentane.
Λ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- Give detailed solution with explanation needed..don't give Handwritten answer....give correct answerarrow_forwardA two-component mixture is analyzed by paper TLC. Component A is more soluble in the mobile phase than component B. The following result is obtained. Calculate the R for each component and label the identity of each spot. 2 Prelaboratory Assignment for Paper Chromatography 0 -Sporting an on 1 by garrow_forwardhow does all this concepts relate to each other. Molarity, solute, solvent, Absorbance, Spectroscopy, and Law Lambert and Beer.arrow_forward
- Kindly answer the question number 2 ONLY based on the objectives of the activity and the question of the activity.arrow_forwardGive correct detailed Solution with explanation needed..don't give Handwritten answer..don't use Ai for answering this..explain all rulesarrow_forwardKindly please help me to answer this accurately. help...arrow_forward
- Answers for sub items D and E.arrow_forwardGive correct detailed Solution with explanation needed with structures...don't give Handwritten answerarrow_forwardChange the bond between the two carbon atoms in each molecule to a double or triple bond as needed to complete the structure. If the bond should remain a single bond, then you do not need to do anything to the bond. Do not change any other bonds in the molecules. Molecule A Molecule B Select Draw Rings More Erase Select Draw Rings More Erase // C H. H. H. H. H H. H. H. H. H. MocRook Proarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning