
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Transcribed Image Text:Propert es Rev x
E Pypor Fulkers x
E Pypor Fulkers x
O Mal -Fukersx Thomas, Gebri x
nent/d/1gQ-IsBbucwMOGIVPUnbTx13fvoW_HIOYBH70iemw3bw/edit
view #2
☆回
Is Add-ons Help Accessibility
Last edit was 2 minutes ago
Courior New -
- 12 +
BIUA
田回-
=- 三 : =-E E
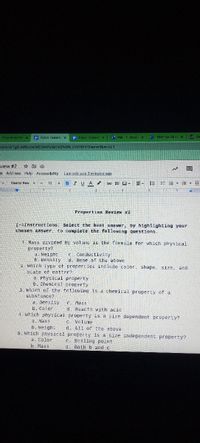
Properties Review #2
T-=Instructions: Select the best answer, by highlighting your
chosen answer, to complete the following questions.
1. Mass divided by volume is the formula for which physical
property?
a. Weight
b. Density
which tYpe of properties include color, shape, size, and
e Conductivity
d. None of the above
state of matter?
a Physical property
6. Chemical property
3. which of the following is a chemical property of a
substance?
a. Densily
b. Color
4. Which physical property is a size dependent property?
a. Mass
b. Weight
5. which physical property is a size independent property?
a. Color
b. Mass
C. Mass
d. Reacts with acid
C. Volume
d. AIl of the above
c. Boiling point
d. Both b and c
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 3. Which of the following states of matter has a fixed volume and a fixed shape? a. Liquid b. Solid C. Gas d. Solution e. Heterogeneous mixture O a b O c d. earrow_forward10. When an irregularly shaped solid of mass 105 g was placed in a cylinder that contained 25.00 ml of water, the water level rose to 45.35 ml. What is the density of the solid? A. 0.238 g/mL B. 2.3 g/mL C. 4.3 g/mL D. 5.16 g/ml E. 20.35 g/mLarrow_forward1. Which of the following statements describe chemical property? a. An iron bar rusts. b. Ice melts when heated. c. The density of iron is 7.87 g/cm3 d. Aluminum is a silver-colored metal. 2. Which of the following is an extensive property? c. Mass d. Color a. Boiling Point b. Density 3. Measuring the mass and volume of a piece of chalk is a way of investigating its a. Chemical and intensive properties b. Chemical and extensive properties c. Physical and intensive properties d. Physical and extensive propertiesarrow_forward
- 7. A student combines pure NaCl (sodium chloride) with pure DI water. The resulting is an example of: a. homogeneous mixture b. heterogeneous mixture c. pure substance d. suspensionarrow_forwardClassify each statement as an observation, a law, or a theory.a. Chlorine is a highly reactive gas.b. If elements are listed in order of the increasing mass of their atoms, their chemical reactivities follow a repeating pattern.c. Neon is an inert (or non-reactive) gas.d. The reactivity of elements depends on the arrangement of their electrons.arrow_forwardDescribe a method that could be used to separate each mixture.a. iron filings and sand c. the components of inkb. sand and salt d. helium and oxygen gasesarrow_forward
- Gasoline is composed of a variety of different liquid hydrocarbons, which do not separate as time passes. Gasoline is an example of a: A) heterogeneous mixture B) Chemical compound C) Chemical element D) Solutionarrow_forwardWhat area of study in chemistry is concerned with the heat transfers that occur during chemical reactions? b. thermochemistry c. calorimetry a. Stoichiometry The law of conservation of energy states that a. in any chemical or physical change, energy cannot be created or destroyed, only changed in form. b. heat changes occur during chemical and physical changes. c. energy is the capacity to do work or to supply heat d. there are two types of energy, kinetic and potential d. thermodynamicsarrow_forward1. When steam condenses, A. A physical change occurs. B. A chemical change occurs. C. Both physical and chemical changes occur. D. H20 molecules increase in number. E. H20 molecules decrease in number.arrow_forward
- water. What can be done to the mixture to help dissolve the remaining sugar After the mixture is stirred, some sugar crystals remain undissolved in the 12. A recipe calls for 210 grams of sugar to be dissolved in 0.25 liters of water. crystals? Add baking soda Cool it down a. Heat it up с. b. Add more sugar d.arrow_forward6. A piece of solid A has twice the volume of a sample of liquid B. And, B has twice the mass of A. A. Solid A and liquid B have equal densities. B. Solid A has twice the density of Liquid B C. Solid A has 4-times the density Liquid B. D. Liquid B has 4-times the density of Solid A.arrow_forwardA Kool-aid solution has sugar and flavoring as the solute and water as the solvent. If ice cubes of water are added to a pitcher of Kool-aid, what happens to the sugar molecules as the ice melts. A. There is not enough information given to determine this. B. The sugar molecules will react with the ice cubes. C. The number of sugar molecules will decrease and be further apart. D. The number of sugar molecules will remain the same, but will be further apart.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY