
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
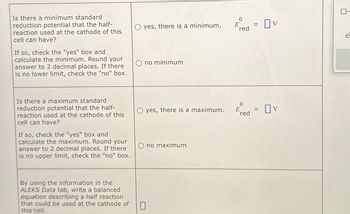
Transcribed Image Text:Is there a minimum standard
reduction potential that the half-
reaction used at the cathode of this
cell can have?
If so, check the "yes" box and
calculate the minimum. Round your
answer to 2 decimal places. If there
is no lower limit, check the "no" box.
0
yes, there is a minimum.
E
=
red
Ono minimum
Ον
e
Ον
Eed = OV
red
Is there a maximum standard
reduction potential that the half-
reaction used at the cathode of this
cell can have?
If so, check the "yes" box and
calculate the maximum. Round your
answer to 2 decimal places. If there
is no upper limit, check the "no" box.
Oyes, there is a maximum.
0
Ono maximum
By using the information in the
ALEKS Data tab, write a balanced
equation describing a half reaction
that could be used at the cathode of
this cell.
☐
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- I need to determine the reduction potentials of all other half-lifes using reduction potential of Zn^2+ (0.10)/Zn half-reaction. I included the data for one of the half-lives I need to find. The known reduction potential for Zn^2+/Zn is -0.79 The cathode is Cu and the anode is Fe The potential (V) for this half cell is 0.767 and it is the potential for the Ecell. The concentration of Cu(NO3)2 is 0.10M and the concentration of Fe(NO)3 is 0.10M In the Cu/Zn pairing the supposed reduction potential of Cu was 0.212. The instructions said to use this value to determine reduction potential of Fearrow_forward0 A certain half-reaction has a standard reduction potential E 'red must provide at least 0.70 V of electrical power. The cell will operate under standard conditions. Note for advanced students: assume the engineer requires this half-reaction to happen at the anode of the cell. Is there a minimum standard reduction potential that the half-reaction used at the cathode of this cell can have? If so, check the "yes" box and calculate the minimum. Round your answer to 2 decimal places. If there is no lower limit, check the "no" box. Is there a maximum standard reduction potential that the half-reaction used at the cathode of this cell can have? If so, check the "yes" box and calculate the maximum. Round your answer to 2 decimal places. If there is no upper limit, check the "no" box. By using the information in the ALEKS Data tab, write a balanced equation describing a half reaction that could be used at the cathode of this cell. Note: write the half reaction as it would actually occur at…arrow_forwardRefer to the galvanic cell below (the contents of each half-cell are written beneath each compartment): V Pt 0.10 M MnO4 0.40 М Cr3+ 0.20 M Mn2+ 0.30 M Cr,0,2- 0.010 M H* 0.010 MH* The standard reduction potentials are as follows: MnO, + 8H* + 5e → Mn2+ + 4H,O, ɛ° = 1.51 V Cr,0,2- + 14H* + 6e 2Cr** + 7H2O, ɛ° = 1.33 V When current is allowed to flow, which species is oxidized? O Cr,0,²- Cr³+ MnO4 Mn2+ O H*arrow_forward
- Help with the following question The 2nd photo is a close uparrow_forwardCalculate Ecell (volts) for the galvanic cell containing the reaction shown below where the concentration of the ion at the anode is 0.500 M and the concentration of the ion at the cathode is 1.50 M. Use the standard reduction potentials from Appendix L in our textbook.arrow_forward7arrow_forward
- A standard galvanic cell is constructed in which a Cd2+ | Cd half cell acts as the cathode. Which of the following statements are correct? Hint: Refer to a table of standard reduction potentials. (Choose all that apply.) In the external circuit, electrons flow from the Cd2+|Cd compartment to the other compartment. As the cell runs, anions will migrate from the Cd2+|Cd compartment to the other compartment. O Cd is oxidized at the cathode. The anode compartment could be I2|I. UThe cathode reaction is Cd2+ + 2e -> Cd.arrow_forwardPlease answer all parts - thank you!arrow_forwardUse smallest possible integer coefficients. If a box is not needed, leave it blank. Use a table of Standard Reduction Potentials to predict if a reaction will occur between Hg metal and I,(s), when the two are brought in contact via standard half-cells in a voltaic cell. If a reaction will occur, write a balanced net ionic equation for the reaction, assuming that the product ions are in aqueous solution. If no reaction will occur, leave all boxes blank.arrow_forward
- please include units in the answerarrow_forwardUse the References to access important values if needed for this question. A standard galvanic cell is constructed in which a Cu²* | Cu half cell acts as the cathode. Which of the following statements are correct? Hint: Refer to a table of standard reduction potentials. (Choose all that apply.) O Cu is oxidized at the cathode. O Cr3+|Cr2+ could be the other standard half cell. The anode reaction could be Cr2* -> Cr³* + e Ag"Ag could be the other standard half cell. U As the cell runs, anions will migrate from the Cu2"|Cu compartment to the other compartment. Submit Answer Try Another Version 2 item attempts remainingarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY