Q: What factor seems to be responsible for the Wagner-Meerwein rearrangement of camphene to give…
A: The rearrangement of camphene to give isobornyl acetate. in general carbocations will rearrange from…
Q: What orbitals (including the unhybridized) are used to form the carbonyl bond in 2- ethylpentanoic…
A: Hybridization is a phenomenon in which the mixing of same or nearly same energy orbital takes place…
Q: PLEASE HELP DRAW AND WRITE THE REACTION, EQUATIONS AND MECHANISMS FOR THE REACTION OF CYCLOHEXENE…
A: Alkene reacts with Br2 in presence of water to give halohydrin.
Q: What is the structure of the principal organic product formed in the reaction of 1-iodopropane with…
A:
Q: Provide the name of the product after heptanoyl chloride reacts with propan-1-ol under acidic…
A:
Q: For the reaction, give the expected substitution product: a) 1-iodo-1-methylcyclohexane +…
A:
Q: State the Conversion of Alcohols to Alkyl Halides with SOCl2 and PBr3 ?
A: Alcohols on reaction with SOCl2 in the presence of pyridine gives Alkyl chloride as the major…
Q: O3 + НО—ССH-CH + CH;CCH,CH(CHO), CO2 но, ?? Zn
A:
Q: Calculate the percentage of 2-chloro-3-methylbutane in the following reaction
A:
Q: Reaction of propene (above) with HBr might, in principle, lead to a mixture of two alkyl bromide…
A:
Q: What will be the major organic product from the dehydration of 2-propanol in the presence of a…
A: Dehydration reaction: This type of reaction is an elimination reaction in which water is formed as a…
Q: Which one of the following alkenes is the Zaitsev product formed by the reaction of hydroxide with…
A: Elimination reactions usually produce the more highly substituted alkene, called the Zaitsev…
Q: During the acid-catalysed hydration of 1-hexene, 2-hexanol is produced. What is the reason for this?
A: Hydration reaction is a type of reaction in which water is combined with a substance. In organic…
Q: what is the product of 3,3-dimethyl-1,5-hexadiene with heat in regards to pericyclic reactions
A: 3,3-dimethyl-1,5-hexadiene undergoes Cope rearrangement in the presence of heat to get the following…
Q: Give the sequence of reactions that produce 2-bromo-4-ethylaniline from aniline. Draw the structures…
A: The molecule 2-bromo-4-ethylaniline contains an amine group which is ortho and para orienting. So,…
Q: Give the structures of the substitution products expected when 1-bromohexane reacts with(a) NaOCH2CH
A:
Q: 1. PPH3 Br X reacts further with carbonyl- containing compound (not part of this question) 2. MeLi…
A: Alkyl halide reacts with PPh3 to give a phosphorous derivative. This on reaction with strong base,…
Q: A small amount of 4,6-decadiyne is formed in reactions involving 1-pentyne. At what point in the…
A: To Find: To find at which point in the mechanism does 4,6-decadiyne is formed.
Q: Ilustrating with an equation show how oxirane can be converted to 1,2 -diphenoxyethane
A: The conversion from oxirane to 1,2, -diphenoxyethane is shown as,
Q: Addition of HCl to 3-methyl-1-pentene gives two products. One of these is 2-chloro-3-methylpentane.…
A:
Q: What reaction could occur with 2-chloro-2-methylpropane when it is mixed with a solution of sodium…
A: The reaction that occurs between 2-chloro-2-methylpropane and a solution of sodium iodide in acetone…
Q: What is the major product of nitration of p-tert-butylmethylbenzene reaction?
A: Given reaction,
Q: Write down the reaction mechanism for the electrophile addition of water to 1-pentene in the…
A:
Q: What would be the major product formed from the reaction of 1-phenyl-2- butene with NBS?
A: In this question we have to tell the major product of the reaction.
Q: Consider an acid-catalyzed dehydration reaction of 2-methyl-2-pentanol. Which product(s) would be…
A: In acid catalytic dehydration reaction the the water molecule eliminate from adjacent carbon atoms…
Q: What products would be obtained from the reaction of 1,3,5-hexatriene with one equivalent of HBr?…
A:
Q: An isomer of tetramethylbenzene undergoes nitration to yield a single product. Based on this…
A: An isomer of tetramethyl benzene undergoes nitration to yield a single product. Based on this…
Q: 1-butanol is reacted with PCC and carried out with an anhydrous solvent DCM, what is the…
A: PCC can oxidize the alcohol.
Q: what alkyl halide will hexane form if it undergoes free radical subsitution?
A: Free Radical Substitution:- In this type of substitution, the saturated(single) bonds undergo…
Q: Provide the structure of the carbene that results when diazomethane decomposes.
A: Provide a diazomethane is decomposed of carbene structure. Diazomethane is a heat it's break of…
Q: ) Specifically, what is the difference between the cyclopentadienyl anion and pyrrole?
A: Pyrrole is an organic compound with the molecular formula C4H4NH and cyclopentadienyl anion is also…
Q: Which of the following will be the major product of the substitution reaction of…
A: Answer :- ---------------------------------------------------------------------------------
Q: What will be the major organic product from the dehydration of 2-hexanol in the presence of a strong…
A: Dehydration of alcohol is the elimination of water to form alkene. At high temperature, more…
Q: Which of the following is expected to be the major organic product when 2-methylbutane is allowed to…
A: Here we have to show the major product of the reaction of 2-methylbutane with Br2 in presence of…
Q: What products will be obtained mainly by nitration of 2-methylbutane? Write a reaction scheme and…
A: 2-methylbutane is an alkane and alkanes are very less reactive compounds. Their less reactivity is…
Q: HO
A: - Provide all of the steps and reagents required for the following chemical conversion. More than…
Q: Write the structure of the product that may result from the dehydrobromination of 2,…
A: Dehydrobromination of haloalkane forms two alkene out of which is minor product and one is major…
Q: What is the most stable radical that could form from 2,2-dimethylbutane and the structure of the…
A:
Q: Write the product of the following reaction. Indicate the mechanism of the reaction involved.
A: A question based on Gilmann reaction, which is to be accomplished.
Q: Show how the o-bromopropylbenzene can be synthesized from benzene:
A:
Q: Which of the following pair of reactions were used in the synthesis of 3-chloro-6-popyllbenzoic acid…
A: Given: To synthesis 3-chloro-6-popyllbenzoic acid from benzene.
Q: What is the set of reagents in order to change 3-methyl-1-butyne into…
A: In this question, we will see the set of Reagents for this transformation. You can see details…
Q: the complete hydrogenation of 2,4-octadiyne will result in what product?
A: Hydrogenation of an unsaturated hydrocarbon gives a saturated hydrocarbon.
Q: Describe a sequence of reactions by which 1,2-dichlorobutane could be prepared from ethyne.
A: Ethyne is a compound which is made up of two carbons while 1,2-dichlorobutane is made up of four…
Q: The reaction of 1,2-dichlorohexane with excess sodium amide in presence of heat produced
A: Sodium amide is an excellent base. It causes double dehydrohalogenation reactions when treated with…
Q: The reaction of 1−methylcyclohexene with peroxyacetic acid will produce a racemic mixture of the…
A:
Q: What are the products prepared for preparation of p-Iodonitrobenzene?
A: Halogen is ortho-para directing group of benzene.
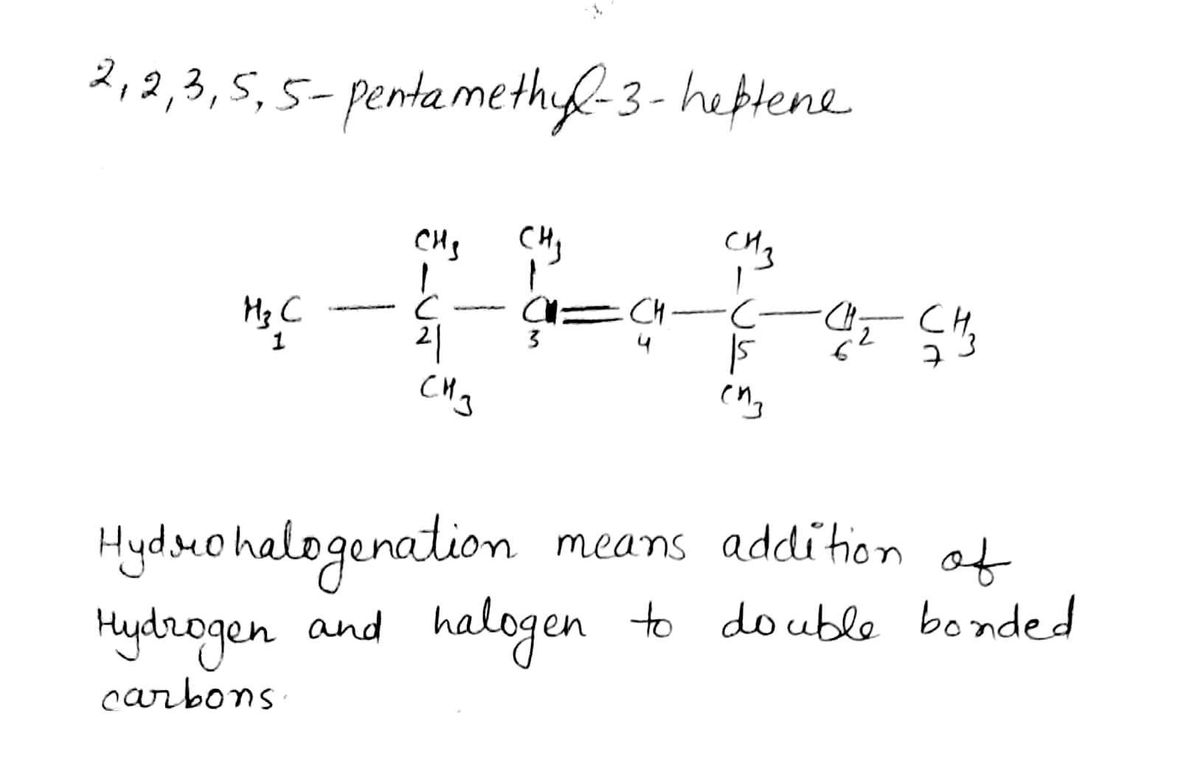
in the hydrohalogenation reaction of 2,2,3,5,5-pentamethyl-3-heptene with HCL, what is the minor product formed?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- The acid-catalyzed dehydration of 2,3-dimethyl-3-pentanol yields three alkene products. What are the names of the three alkenes? Which of the three alkenes is the major product?What is the structure of the principal organic product formed in the reaction of 1-iodopropane with NaOH?When bromine is added to two beakers, one containing phenyl isopropyl ether and the other containing cyclohexene, the bromine color in both beakers disappears. What observation could you make while performing this test that would allow you to distinguish the alkene from the aryl ether?
- Lipoic acid is required by many microorganisms for proper growth. As a disulfide, it functions in the living system by catalyzing certain oxidation reactions and is reduced in the process. Write the structure of the reduction product.Write a series of reactions leading to para-bromoethylbenzene, beginning with benzene and using other reagents as needed. What isomeric side products might also be formed?What product is formed when the mixture of methyl-2-bromine-2-butane with isopropyl bromide and metallic sodium is heated? Write the chemical equation of the reaction
- Alkyl sulfonates undergo the same type of substitution reactions as alkyl halides and can also be prepared from alcohols. What advantage does the preparation of an alkyl sulfonate from an alcohol have over the preparation of an alkyl halide from an alcohol?a) What products would you expect from the elimination reaction of 3-Bromo-2- methylpentane? Show the reaction by writing the condensed structural formula of the reactants and products. Identify the major and minor products. b) What alkyl halide might the 3,6-Dimethyl-1- heptene have been made from?1) The acid-catalyzed dehydration of 2-methyl-2-butanol yields two alkene products, what are the names of the two alkenes? 2) which of the two alkenes is the major product?
- Why does the reaction of t-butanol with sulfuric acid gives the elimination product isobutylene whereas the reaction with HCl gives predominantly the substitution product?The condensed structural formula of an organic compound is CH3-CH2-CHBr-CHCl-CHCl-CHO. What is the name and type of this organic compound? 3-bromo-4,5-dichloro hexanal; aldehyde 4-bromo-2,3-dichloro hexanal; aldehyde 4-bromo-2,3-dichloro hexan-1-one; ketone 3-bromo-4,5-dichloro hexan-1-one; ketoneAn organic compound S reacted with ozone to give compounds Q and R. Both Q and R react with NaOH/I2 to produce a yellow precipitate with antiseptic smell. Q reacts with HCN to produce the compound CH3C(CH3)OHCN while R gives a silver mirror with Tollens reagent. I) write the structural formulae and names of Q,R,S and the Yellow Precipitate. II) write equations to show the reactions Q with NaOH/I2 and R with H^+/Cr2O7^2-

