
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Could you help me with this question? I don't know where to start. All the information has been provided.
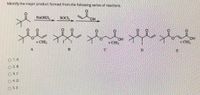
Transcribed Image Text:Identify the major product formed from the following series of reactions.
NaOHЛ,
SOCI,
+ CHI3
HO.
+ CHI
HO.
+ CHI,
A
B.
C.
D
E
O 1.A
O 2. B
O 3. C
O4. D
5. E
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the mass (in kg) of 6.89 × 10^25 molecules of carbon dioxide? Give the answer as a number. The units are understood to be kg.arrow_forwardA student prepares a 0.38 mM aqueous solution of acetic acid (CH3CO₂H). Calculate the fraction of acetic acid that is in the dissociated form in his solution. Express your answer as a percentage. You will probably find some useful data in the ALEKS Data resource. Round your answer to 2 significant digits. 11 % 0.0 x10 X S 0 Earrow_forwardAssuming that the copper and lead are pure, determine the relative amounts of each kind of BB. The density of copper is 8.96 g/cm ^ 3 . The density of lead is 11.4 g/cm ^ 3arrow_forward
- Ethanol has a density of 0.789 g cm‐3. Calculate the volume in mL that must be poured into a measuring cylinder to give 19.8 g of ethanol. Note 1 mL = 1 cm3.arrow_forwardA hypothetical metal has a theoretical density of 11.47 g/cm3. This metal adopts a cubic crystal structure with an edge length of 0.387 nm and atomic radius of 0.137 nm. Find the atomic weight of this metal in g/mol. NA = 6.022 x 1023 atoms/mol. Express your answer in two decimal places only. Do not put the units.arrow_forwardSand and iron particles that are similar in size and color are mixed together in a beaker. What would be the best method of separating the particles? A- Add water to the mixture B- Use tweezers to separate them. C- Pour the mixture into a filter. Use a magnet to separate them.arrow_forward
- Which piece of equipment would you use to measure 7.10 mL of CuSO4 (aq)?arrow_forwardA chemistry student weighs out 0.112 g of formic acid (HCHO2) into a 250. mL volumetric flask and dilutes to the mark with distilled water. He plans to titrate the acid with 0.0800M NaOH solution. Calculate the volume of NaOH solution the student will need to add to reach the equivalence point. Be sure your answer has the correct number of significant digits.arrow_forwardPart H Write balanced net ionic equation for NazPO4(aq)+ CaCl2 (aq) → Ca3(PO4)2(s)+ NaCl(aq). Express your answer as a chemical equation. Identify all of the phases in your answer.arrow_forward
- The chemistry teacher needs a solution made up of a 3 g sugar cube (C6H1206) dissolved in a 250 ml teacup. Find the molarity of this solution. Be sure to report your answer to the correct number of significant figures. Do not include units.arrow_forwardThe world population is estimated to be 7.4 x 109. Nauru is the smallest island nation and comprises 1.5 x 10-4% of the world population. If the percentage of left-handed people is approximately 12%, estimate the number of left-handers on the island of Nauru.arrow_forwardThe chemical formula for hydrogen bromide is HBr. A chemist measured the amount of hydrogen bromide produced during an experiment. She finds that 5.9 g of hydrogen bromide is produced. Calculate the number of moles of hydrogen bromide produced. Be sure your answer has the correct number of significant digits. 477. mol x10 X S 4arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY