
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
I am having trouble finding the rest of the data for my precipitation Lab for percent yield of cobalt phosphate. The data I collected has already been imputed but cannot find the rest of the data. I need to use the given data I collected to find the other blank boxes in excel.
![### Precipitation Reactions – Background
This section provides instructions on how to calculate various aspects of a precipitation reaction.
#### A. Calculate the Mass of Dried Precipitate
#### B. Calculate the Moles of Na₃PO₄ Used
\[ \text{moles Na}_3\text{PO}_4 = (\text{molarity of Na}_3\text{PO}_4, \text{mole/L}) \times \left(\frac{\text{volume of Na}_3\text{PO}_4, \text{mL}}{1000}\right) \]
#### C. Calculate the Moles of Co(NO₃)₂ Used
\[ \text{moles Co(NO}_3\text{)}_2 = (\text{molarity of Co(NO}_3\text{)}_2, \text{mole/L}) \times \left(\frac{\text{volume of Co(NO}_3\text{)}_2, \text{mL}}{1000}\right) \]
#### D. Using the Balanced Chemical Equation, Convert the Moles of Na₃PO₄ to Moles of Precipitate
\[ \text{moles Na}_3\text{PO}_4 \times \left(\frac{\text{stoich. coeff. precipitate}}{\text{stoich. coeff. Na}_3\text{PO}_4}\right) = \text{moles of precipitate} \]
#### E. Using the Balanced Chemical Equation, Convert the Moles of Co(NO₃)₂ to Moles of Precipitate
\[ \text{moles Co(NO}_3\text{)}_2 \times \left(\frac{\text{stoich. coeff. precipitate}}{\text{stoich. coeff. Co(NO}_3\text{)}_2}\right) = \text{moles of precipitate} \]
#### F. What is the Limiting Reagent and What is the Theoretical Yield (Moles) of Precipitate?
#### G. Calculate the Molar Mass of the Precipitate
**Note:** The precipitate formed in this experiment is actually a hydrate!
**Precipitate \cdot X H₂O**
Perform literature research to determine the most common hydrate of the precipitate. Based on your answer, calculate the correct molar mass](https://content.bartleby.com/qna-images/question/cf266520-407e-4b51-839c-e1d92d759be6/9d93c7c2-64e5-4168-993d-7ae5af2c435d/ulc576_thumbnail.jpeg)
Transcribed Image Text:### Precipitation Reactions – Background
This section provides instructions on how to calculate various aspects of a precipitation reaction.
#### A. Calculate the Mass of Dried Precipitate
#### B. Calculate the Moles of Na₃PO₄ Used
\[ \text{moles Na}_3\text{PO}_4 = (\text{molarity of Na}_3\text{PO}_4, \text{mole/L}) \times \left(\frac{\text{volume of Na}_3\text{PO}_4, \text{mL}}{1000}\right) \]
#### C. Calculate the Moles of Co(NO₃)₂ Used
\[ \text{moles Co(NO}_3\text{)}_2 = (\text{molarity of Co(NO}_3\text{)}_2, \text{mole/L}) \times \left(\frac{\text{volume of Co(NO}_3\text{)}_2, \text{mL}}{1000}\right) \]
#### D. Using the Balanced Chemical Equation, Convert the Moles of Na₃PO₄ to Moles of Precipitate
\[ \text{moles Na}_3\text{PO}_4 \times \left(\frac{\text{stoich. coeff. precipitate}}{\text{stoich. coeff. Na}_3\text{PO}_4}\right) = \text{moles of precipitate} \]
#### E. Using the Balanced Chemical Equation, Convert the Moles of Co(NO₃)₂ to Moles of Precipitate
\[ \text{moles Co(NO}_3\text{)}_2 \times \left(\frac{\text{stoich. coeff. precipitate}}{\text{stoich. coeff. Co(NO}_3\text{)}_2}\right) = \text{moles of precipitate} \]
#### F. What is the Limiting Reagent and What is the Theoretical Yield (Moles) of Precipitate?
#### G. Calculate the Molar Mass of the Precipitate
**Note:** The precipitate formed in this experiment is actually a hydrate!
**Precipitate \cdot X H₂O**
Perform literature research to determine the most common hydrate of the precipitate. Based on your answer, calculate the correct molar mass
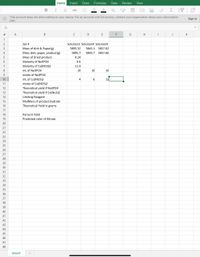
Transcribed Image Text:### Experimental Data Set
#### Mass Measurements
- **Mass of dish & Paper (g)**
- Solution 3: 5895.32
- Solution 4: 5843.1
- Solution 9: 5857.02
- **Mass dish, paper, product (g)**
- Solution 3: 5895.7
- Solution 4: 5843.7
- Solution 9: 5857.66
- **Mass of dried product**
- Solution 3: 0.24
#### Molarity
- **Molarity of Na₃PO₄:** 0.6
- **Molarity of Co(NO₃)₂:** 11.3
#### Volume Measurements
- **mL of Na₃PO₄**
- Solution 3: 10
- Solution 4: 10
- Solution 9: 10
- **mL of Co(NO₃)₂**
- Solution 3: 4
- Solution 4: 6
- Solution 9: 12
#### Calculations
- **Moles of Co(NO₃)₂**
- Solution 9: 4
- **Theoretical yield if Na₃PO₄:** (Not provided)
- **Theoretical yield if Co(NO₃)₂:** (Not provided)
- **Limiting Reagent:** (Not provided)
- **MolMass of product hydrate:** (Not provided)
- **Theoretical Yield in grams:** (Not provided)
#### Results
- **Percent Yield:** (Not provided)
- **Predicted color of filtrate:** (Not provided)
### Notes
The data set outlines the mass of dishes, papers, and products for three solutions, alongside molarity data for two compounds involved in the experiments. Calculated values like moles, theoretical yields, and percent yields require additional input or further calculations.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 9 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- I need help, please answer thank you!arrow_forwardHow many molecules of hydrogen fluoride (HF) are in (2.1x10^-2) moles of hydrogen fluoride (HF)? For Avogadro's number use: 6.022 x 1023 Round your answer to two significant figures. For the unit, use the word molecule. (Please show me how to work. Thank you)arrow_forwardMicrosoft O ift A chemist adds 340.0 mL of a 3.07 mol/L sodium nitrate (NaNO3) solution to a reaction flask. Calculate the mass in grams of sodium nitrate the chemist has added to the flask. Be sure your answer has the correct number of significant digits. W O CHEMICAL REACTIONS Using molarity to find solute mass and solution volume 0 g Microsoft tab Microsoft 6.52.210... esc caps lock 2 ♦ Explanation ! 1 Q A T control option Z Check @ 2 W S #3 X H command x10 E X D NOV 14 $ 4 R S (of F 67 5⁰ % 5 C V T G átv MacBook Pro 6 Y e & 7 H Ⓒ2022 McGraw Hill LLC. All Rights Reserved. U B N 8 J M ( 9 K { 2/5 [ I command option + 11 ? | DOCX 77 } 6arrow_forward
- BACKGROUND KNOWLEDGE: The reaction performed is the precipitation of calcium hydroxide from solutions of calcium chloride and potassium hydroxide. The percentage yield of calcium hydroxide is 98.97% QUESTION: Comment on your calculated percentage yield and the main sources of error in this experiment.arrow_forwardA student prepared benzil according to the experimental procedure using 2.58 g. of benzoin and 12 mL of nitric acid. The mass of the crude benzil product was 2.25 g. and the mass of the recrystallized benzil product was 1.95 g. Find the molecular weights for benzoin and benzil, then answer the following questions. What was the percent yield of the crude benzil? Assume units of %. Round answer to the first decimal place. What was the percent yield of the purified benzil? Assume units of % Round answer to the first decimal place. What was the percent recovery of the purified benzil from the crude benzil? Assume units of % Round answer to the first decimal place.arrow_forward1-4arrow_forward
- When solutions of silver nitrate and sodium chloride are mixed, silver chloride precipitates out of solution according to the equation AGNO3 (aq) + NaCI(aq)→AgCl(s) + NaNO3(aq) Part A What mass of silver chloride can be produced from 1.69 L of a 0.214 solution of silver nitrate? Express your answer with the appropriate units. • View Available Hint(s) ? mass of AgCl = Value Units Submit Part B The reaction described in Part A required 3.48 L of sodium chloride. What is the concentration of this sodium chloride solution? Express your answer with the appropriate units. • View Available Hint(s) HA ? Value Units Submitarrow_forwardA pure sample of an organic molecule has the formula CxHyOz. If x = 11, y = 13, and z = 2. Calculate the percent by mass of oxygen in the formula. Round your result to the nearest tenth number; do not enter a % sign in your answer.arrow_forward1 _ 02>_CO2 +_H20 1)_CsHs +, 6.003g of oxygen, makes how many moles of water?arrow_forward
- If the crucible originally weighs 3.715 g and 2.000 g of hydrate are added to it, what is the weight of the water that is lost if the final weight of the crucible and anhydrous salt is 5.072? Do not include units and assume three decimal places for all numbers. Remember to include the zero before the decimal if the number is less than one.arrow_forwardUse the References to access important values if needed for this question. If 21.2 grams of an aqueous solution of manganese(II) chloride, MnCl, contains 3.65 grams of manganese(II) chloride, what is the percentage by mass of manganese(II) chloride in the solution? % MnCl2 Submit Answer Retry Entire Group 9 more group attempts remaining Previous Nex Say Cengage Learning | Cengage Technical Support 100% 11 近arrow_forwardI have the data, I only need the calculations of the last page.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY