
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Don't use Ai for answering this
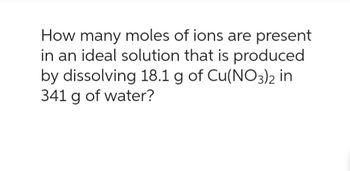
Transcribed Image Text:How many moles of ions are present
in an ideal solution that is produced
by dissolving 18.1 g of Cu(NO3)2 in
341 g of water?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Similar questions
- The triprotic form of the amino acid cysteine is shown below, along with the pK, value for each ionizable site. The isoelectric point (pl) for cysteine occurs at pH= 5.1. B 10.8 CH₂ SH C13 A LA -OH (1) What is the charge on the molecule at the isoelectric point? (2) In the predominant form of the molecule at the isoelectric point, is each of the sites (labeled A, B, and C) protonated or unprotonated? site A site B: site Carrow_forwardAbsorbance = 2.000 - log(%T) If %T = 12.6% what is the absorbance of the solution?arrow_forwardWhat is the Kb for acetate if the Ka for acetic acid is 1.8 x 10^-5 ? Group of answer choices 1. 0.000132 2. 5.56 x 10^-9 3. 5.56 x 10^-10 4. 2.52arrow_forward
- Macmillan Learning Complete the Ksp expression for Ag₂CO3. Ksp = 8.46 × 10-12 = MacBook Pro Search or type URLarrow_forwardpH pH = 4.389 pH = 4.874 = pH = 5.339 pKa Answer Bank Using the equation to calculate the quotient [AT]/[HA] at three different pH values. [A] [HA] + log [A-] [HA] [A-] [HA] = = = LA I [HA] 1 0.3273arrow_forwardWhat is the protien gradient?arrow_forward
- Please answer the following question:arrow_forwardGive a suitable chemical equation to show how the suppressor neutralizes the KOH eluentarrow_forwardWhich absorbance spectrum from the previous problem represents the dye solution with the greatest concentration? 1.4 1.2 Curve 3 0.8 0.6 Curve 2 Curve 1 0.4 0.2 0.2 Wavelength (nm) O All three are approximately the same concentration O Curve 3 O Cannot be determined from the information given Curve 2 Curve 1 700 580 Absorbancearrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY