
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
help please #1
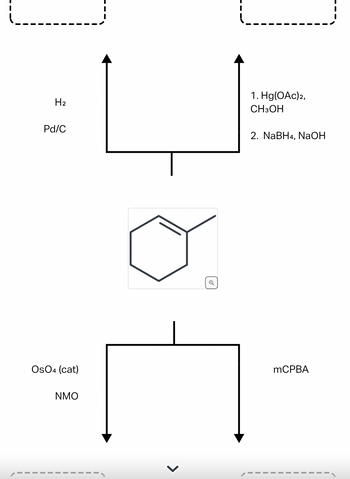
Transcribed Image Text:H₂
Pd/C
OSO4 (cat)
NMO
1. Hg(OAc)2,
CH3OH
2. NaBH4, NaOH
mCPBA

Transcribed Image Text:A common alkene starting material is shown below. Predict the
major product for each reaction. Use a dash or wedge bond to
indicate relative stereochemistry of substituents on
asymmetric centers, where applicable. Ignore any inorganic
byproducts
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 5. Suppose you have a mixture of water and your 2-chloro-2-methylbutane product in a separatory funnel. Use densities to predict which phase will be the top layer in the funnel. a. 2-chloro-2-methylbutane (organic phase) b. water (aqueous phase) c. there would only be one phase since the substances are misciblearrow_forwardA sample of water has the following concentration of ions as shown in the attached table (and the pH is near neutral). What is the total hardness (TH)? What is the carbonate hardness (CH)? What is the noncarbonate hardness (NCH)?arrow_forwardHow long (in seconds) does it take for the concentration of A to decrease to 0.00800 M? Enter your answer with one decimal place and no units. 1/[A] 120 100 80 60 40 20 0 0 10 20 y = 0.7957x + 61.714 R² = 0.9998 30 Time (sec) 40 50 60arrow_forward
- PLEASE SHOW YOUR WORK (THIS IS NOT A GRADED ASSIGNMENT)arrow_forward50 mL of stock solution were taken and added to flask. Then 50 mL of Di water were added to the flask. This is solution 1. Then 50mL of solution 1 were taken and added to a flask and 50 mL of Di water were added to the flask. This is solution 2. Then 50mL of solution 2 were taken and added to a flask and 50 mL of Di water were added to the flask. This is solution 3. Then 50mL of solution 3 were taken and added to a flask and 50 mL of Di water were added to the flask. This is solution 4. Find the concetrations of each solution. Information of stock solution - molar mass- 534.3g/mole 0.587g in MilliQarrow_forwardUse the following information to answer numerical-response question 10. Crude oil is separated into different groups of hydrocarbons, or fractions, in a fractional distillation tower. Two different locations at which fractions can be removed from the tower are labelled in the diagram below. Numerical Response Fractional Distillation Tower Crude oil K-11090 the greatest amount of CH4(g) is numbered the largest molecules is numbered molecules with the highest boiling points is numbered 10. Match the numbers of the locations in the diagram above with the descriptions of the fractions that could be removed as given below. The location with the fraction that contains molecules that are least attracted to one another due to weak intermolecular forces is numbered Location 1 190071 Location 2 zonirabi woll (Record in the first column) (Record in the second column) (Record in the third column) (Record your answer in the numerical-response section on the answer sheet.) (Record in the fourth…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY