
Biochemistry
9th Edition
ISBN: 9781319114671
Author: Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher: W. H. Freeman
expand_more
expand_more
format_list_bulleted
Question
thumb_up100%
question 28 // both pictures are attached below.
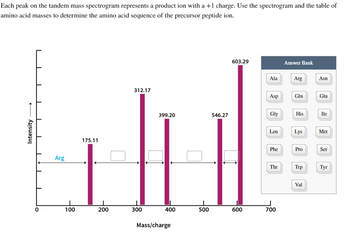
Transcribed Image Text:Each peak on the tandem mass spectrogram represents a product ion with a +1 charge. Use the spectrogram and the table of
amino acid masses to determine the amino acid sequence of the precursor peptide ion.
4
Intensity
I
Arg
100
175.11
200
312.17
300
399.20
400
Mass/charge
O
500
546.27
603.29
600
Ala
Asp
Gly
Leu
Phe
Thr
700
Answer Bank
Arg
Gin
His
Lys
Pro
Trp
Val
Asn
Glu
Ile
Met
Ser
Tyr
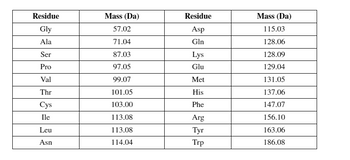
Transcribed Image Text:Residue
Gly
Ala
Ser
Pro
Val
Thr
Cys
Ile
Leu
Asn
Mass (Da)
57.02
71.04
87.03
97.05
99.07
101.05
103.00
113.08
113.08
114.04
Residue
Asp
Gln
Lys
Glu
Met
His
Phe
Arg
Tyr
Trp
Mass (Da)
115.03
128.06
128.09
129.04
131.05
137.06
147.07
156.10
163.06
186.08
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps

Knowledge Booster
Similar questions
- QUESTION 30 Figure 1.21C Computed topography scan. Identify the plane of section of the medical imaging scan in Figure 1.21C A. Transverse plane B. Midsaggital plane C. Frontal plane O D. Parasagittal planearrow_forwardQUESTION 13 The organism represented in the above image is in the genus Euglena. is a flagellate. is in the genus Amoeba. makes pseudopodia. is an excavate. is a chromalveolate. Copyright © McGraw-Hill Education. Permission required for reproduction or display O Kage Mikrofotograffie/Phototakearrow_forwardQuestion 69SavedA client is having electroconvulsive therapy for the first time. What potential risk exists with this type of therapy? Question 69 options: hearing impairment generalized seizure activity retrograde amnesia frontal lobe atrophyarrow_forward
- QUESTION 13 RNA is pretty easy to make into DNA because it's single stranded, doesn't form as many structures, and is not as easily degraded. O True O Falsearrow_forwardQuestion 128 Shingles is reactivation of A person suffering from shingles is O polio; shedding virus in feces HPV; shedding virus in sloughed stratified squamous keratinized epithelial cells of skin O Varicella/chickenpox virus; shedding infectious virus from lesions until healed HIV: shedding virus in bloodarrow_forwardQuestion 35arrow_forward
- Please answer very soonarrow_forwardQuestion 15 general , the mouse receiving the PTU in his / her food: 1. consumed more oxygen at all temperatures 2. ate more than the control mouse 3. consumed less oxygen at all temperatures 4. produced more oxygen at all temperaturesarrow_forwardQuestion 5 Chlamydia O symptoms of infection appear within 1-2 days after exposure. is best prevented with hormonal birth control. can cause infertility if left untreated both in men and women. O can cause infertility if left untreated only in men.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON