Q: dont provide handwriting solution....
A: The portions of 1,2,3-Trichloropropane that interact differentially with water molecules are…
Q: Predict the structure of the alkene you would use to prepare the alkyl halide below. Br (only…
A: In case of any doubt please feel free to ask.
Q: Draw the reaction mechanism of 4-aminobenzenesulfonic acid with 2-naphthol for the formation of an…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Change Add water Add oxygen Lower the temperature Raise the temperature Decrease the pressure 2H2(g)…
A: Step 1:Given reactions,1st ) exothermic , having more moles of Gas at reactant side 2nd )…
Q: D. Make Pt(H2O)2Br2Cl2. If this molecule has isomers (geometric and/or optical) draw all of them.
A: Step 1: The given compound [PtBr2Cl2(H2O)2] is an octahedral compound.The name of the compound…
Q: a segment of dna has the following sequences of bases ATGCAATGATATTGAAGCTTA
A: The DNA segment you've provided, "ATGCAATGATATTGAAGCTTA," is composed of nucleotide bases. These…
Q: In an order 1 kinetic reaction, I have the following data table: t/ °C 300 0,00004 200 0,004 How…
A: The equation of half-life for first order reaction is:The rate constant (k) is highly dependent on…
Q: None
A: Boyle's law is a gas law, stating that the pressure and volume of a gas have an inverse…
Q: The standard cell potential (E°cell) for the reaction below is +0.63 V. The value of ΔG° for the…
A: Step 1: The relationship between cell potential (E°cell)and Gibbs energy (ΔGo) can be expressed by…
Q: None
A:
Q: Prepare the compound by using suitable reagents. More than one step will likely be required.
A:
Q: What is the relationship between the structures shown? OH and OH HO Select one: OA diastereomers B.…
A: 1.Structure (I) and (II) have molecular formula and same arrangment of atoms, they differ only in…
Q: None
A: The difference in boiling points between carbon tetrachloride (CCl4) and chloroform (CHCl3) can be…
Q: What would the potential of a standard hydrogen electrode (S.H.E.) be under the following…
A: Here's how to find the potential of the SHE under these conditions:Standard Hydrogen Electrode…
Q: Complete solutions need
A:
Q: In my Chemistry textbook (Chemistry: The Central Science), it says:Step 1: NO(g) + Br(2)(g)…
A: -------I m sure this will help you I hope you will appreciate my efforts Thank you!!!!!Happy…
Q: Consider the following system at equilibrium where AH = 182 kJ and K = 7.00 x 10-5, at 673 K.…
A: First Question.) a.) FALSE. The enthalpy change (ΔH) of the reaction is positive, which indicates…
Q: ATM Organic X My Cou X Macmill × M Inbox (1 × M ZOOM X 6 ← → C app.aktiv.com M Gmail YouTube ATM…
A:
Q: balance this equation : HF(aq) + LiOH (aq)
A:
Q: Check ALL of the statements below that are true when a strong acid is titrated by slowly adding…
A: Step 1: A. It is true that before any base is added the pH of the solution will be acidic . As the…
Q: How many organic products are there in the reaction of 1,3-cyclohexadiene with HBr? (note: be sure…
A:
Q: Imagine the main chain of a protein bends back on itself, so that two amino acid residues R₁ and R₂…
A: Can the amino acid residue R₁ form a hydrogen bond with residue R₂ at physiological pH, if R₂ were…
Q: What product(s) result from the Claisen condensation carried out with an equimolar mixture of ethyl…
A: Step 1:
Q: What changes can be made to the hydrogenation reaction to make it safer for beginner students?
A: Based on the information which you have provided in question, which depicts a step in the transfer…
Q: Chemistry
A:
Q: Please don't provide handwritten solution.
A: PThe prompt asks you to:Draw all reasonable elimination products to the right of the arrow.Redraw in…
Q: :$;$;$;$$:$$:$;$
A: Step 1: Step 2: Step 3: Step 4:
Q: if i didn’t pay attention to which bottle I grabbed and accidentally used hexane instead ofethyl…
A: For your reference, the order of elution will be based on what solvent is used in the mobile phase.…
Q: S. Don't provide handwriting solution
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Part a through c please a) You are presented with 12.0 kilograms (kg) of sugar for a rum brewing…
A: Part a)a) 423. ozHere's how to find the number of ounces of sugar in 12.0 kg:Convert kilograms (kg)…
Q: The following reaction involves an CI A O a. intermolecular SN1 reaction. O b. intramolecular SN2…
A: Step 1:Q.3 option d not clear in image and options d is correct if this question contains four…
Q: Name these organic compounds please:
A: Step 1: Notice that all the given compounds only have single bonds. This means that all three…
Q: 1. Why is the soap translucent? 2. What helps the soap retain its water content? 3. Draw a…
A: Figure showing the soap molecules. parts of gold from japand diamonds. REFERENCE: 5.2: Soap -…
Q: ;$;$;$):):$:
A: For question 2 SN2 reaction. this is cyclohexane, chloro cyclohexane reacting with ammonia which is…
Q: Using the blank graph space below, draw an energy diagram/profile for a reaction that includes the…
A:
Q: The rate constant of a practically irreversible second-order reaction 2 A → B has a value of 0.145…
A: In solving this problem, integrated rate law for a second-order reaction is used. The integrated…
Q: slove part c
A: Sure, The temperature rise, standard molar enthalpy of formation, and C-C bond dissociation energy…
Q: Decide whether these proposed Lewis structures are reasonable. proposed Lewis structure Is the…
A: Step 1: Step 2:
Q: Draw the shape, hybridization and expected-1 angles in the following a- XeО,F b- IF3 c- Br PCl₂ d-…
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw the major organic product of the following reaction. + PC13
A:
Q: Please answer 29
A: In case of any doubt please feel free to ask.
Q: None
A: Condensed Structural Formula:The condensed formula provides a simplified representation of the…
Q: :$;$;););):$::$:&&:$$;$;;$
A: Step 1: Information: As their are different types of reaction performed with alkyne gives diffrent…
Q: No need to upload any image give me the name with proper explanation will upvote.
A: The Foundation: The Carbonyl Group (C=O)At the heart of methyl isopropyl ketone lies the carbonyl…
Q: Write down the Lewis dot structures of each of the following molecules. Check the formal charge in…
A: Now let's draw the Lewis dot structure for each molecule givena) CH2Cl2 In this Lewis structure,…
Q: Chemistry
A: Step 1:Step 2:
Q: Chemistry
A: Recrystallization is based on the principal of solubility of any compound in a particular solvent at…
Q: QUESTION 1 Combustion of 7.67 g of liquid benzene (C6H6) causes a temperature rise of 47.6 °C in a…
A: Step 1: Calculate the heat absorbed by the calorimeter. qcal = Ccal ΔTwhereCcal = heat capacity of…
Q: 12. Which solution has the highest hydronium ion concentration? (A) 0.10 M NaOH (B) 0.02 M CH3COOH…
A: Note that: Hydronium ion (H3O+ or H+) concentration is a measure of the acidity of a solution, with…
Q: Solution for the uploaded question please.
A: Step 1:Step 2:Step 3:
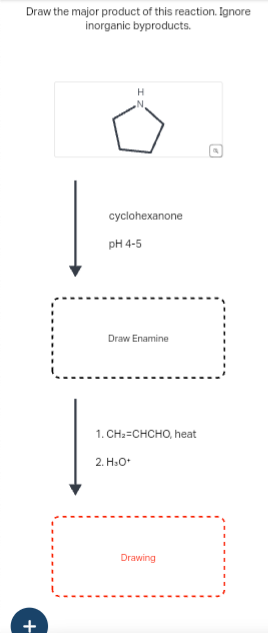
Draw the major product of this reaction. Ignore inorganic byproducts.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Which reagents would you use to perform reaction B?a) PBr3.b) p-toluenesulfonyl chloride.c) SOCl2.d) NaOCl, NaOHDraw the products of each step of this reaction sequence. Ignore inorganic byproducts.help me draw the products when ethyl isopropyl ether is treated with the following reagent: 1.HBr (2 equivalents) 2.HI (in excess)
- Draw the major product of this reaction. Ignore inorganic byproducts and CO2.help me draw the products when 3-isopropylhexan-2-ol is treated with the following reagents: 4. SOCl2, pyridine 5. PBr3 6. TsCl, pyridine 7. [1] NaH; [2] CH3CH2Br 8. [1] TsCl, pyridine; [2] NaSH 9. POCl3, pyridine help me draw the products when propylene oxide is treated with the following reagent: 4. [1] CH3S-; [2] H2O 5. [1] OH-; [2] H2OWhat product is formed when CH3OCH2CH2C ≡ CCH2CH(CH3)2 is treated with each reagent: (a) H2 (excess), Pd-C; (b) H2(1 equiv), Lindlar catalyst; (c) H2 (excess), Lindlar catalyst; (d) Na, NH3?
- Chemistry Draw the product formed when phenylacetaldehyde (C6H5CH2CHO) is treated with reach reagent: f. (CH3)2CHNH2, mild acid g. (CH3CH2)2NH, mild acid h. CH3CH2OH (excess), H+ i. piperidine, mild acid j. HOCH2CH2OH, H+18. Show an efficient synthesis of 2-methyl-1-cyclohexanol from 1-methylcyclohexene. Show the structure of all reactants and products.draw the major product of this reaction. ignore inorganic byproducts and phthalhydrazide side-product nh2nh2 koh

