
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
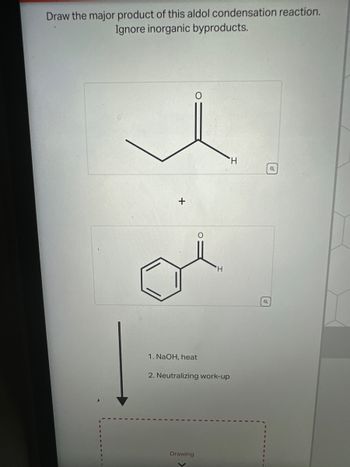
Transcribed Image Text:Draw the major product of this aldol condensation reaction.
Ignore inorganic byproducts.
+
H
H
1. NaOH, heat
2. Neutralizing work-up
Drawing
く
a
a
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the major product of this aldol addition reaction. Ignore inorganic byproducts. 1. LDA 2. C6H11CH2CHO 3. Neutralizing work-up Drawing Qarrow_forwardPlease don't provide handwriting solutionarrow_forwardDraw the major product of this aldol addition reaction. Ignore inorganic byproducts. 1. LDA 2. C6H11CH2CHO 3. Neutralizing work-up Select to Drawarrow_forward
- Draw the major product of this aldol condensation reaction. Ignore inorganic byproducts. + H a H Q 1. NaOH, heat 2. Neutralizing work-up OH Tap here for additional resourcesarrow_forwardDraw the major product of each step in this reaction sequence. Ignore inorganic byproducts. Select to Draw OH (CH3)3CSI(C H3)2CI, Et3N Bu4NF (TBAF) Select to Draw 1. CH3MgBr 2. H3O+ Select to Drawarrow_forwardDraw the major product of this aldol condensation reaction. Ignore inorganic byproducts. 1. NaOH, heat 2. Neutralizing work-up + H H ☑arrow_forward
- Draw the major product of each step in this reaction sequence. Ignore inorganic byproducts. HO Select to Draw (CH3)3SICI, Et3N HCI, H₂O Select to Edit NaBH4 CH3OH Select to Drawarrow_forwardDraw the major product of each step in this reaction sequence. Ignore inorganic byproducts. Select to Draw OH (CH3CH2) 3SICI, Et3N LiF Select to Draw 1. I 2. H3O+ $1 CH3CH2MgBr Select to Drawarrow_forwardDraw the major product of this aldol addition reaction. Ignore inorganic byproducts. 1. LDA 2. CoHnCHzCHO 3. Neutralizing work-up Drawing Qarrow_forward
- ■ Draw the major product of the reaction sequence. Omit byproducts. 1. PhCOOOH 2. EtLi, then H3O+ 3. PCC Select Draw C Rings H O More Erase Q2 Qarrow_forwardDraw the major product of this solvolysis reaction. Ignore any inorganic byproducts.arrow_forwardDraw the major products of this reactionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning