Draw one of the two enantiomers of the major product from this reaction. Use wedge and dash bonds to indicate stereochemistry where appropriate. Ignore inorganic byproducts. Br2
Q: Is the name correct? 5-(2,2-dimethylethyl)nonane If not, give the correct name.
A:
Q: %3D and cis trans Explain why cis-bicyclo[4.1.0]heptane is more stable than its trans isomer. Answer...
A: The solution of the answer is given below:
Q: Imagine there is a carbon reservoir that holds 12,500 GtC. The flux out of this reservoir is 50 Gt/C...
A: Please find your solution below : Turn over time is defined as the time taken to fill or empty the p...
Q: Which element is the most metallic? F Au Fr Li What helps particles move? Kinetic Molecular So...
A: First group elements are most metallic character. so lithium (Li) is correct option. K...
Q: 6. Idantify the orerapping orbitalou im volvet i the formation of cach Yum bered bond. NO. HO-C-H ČH...
A: We have to tell the overlapping orbitals involved in formation of each number of bonds in the below ...
Q: There is a barrier to rotation in alicyclic alkanes because of steric strain. Select the correct res...
A:
Q: Substances A and B are two miscible liquids that behave as an ideal mixture. Use the following liqui...
A: Since you have posted multiple questions, we are entitled to answer the first only. The temperature ...
Q: Which of the following is NOT true of electrons? Group of answer choices 1. they have a negative ch...
A:
Q: What is the dilution factor needed to change a 1:2 dilution to a 1:10?
A: To change 1:2 dilution to a 1:10, we have to dilute solution by a factor of 5.
Q: 1. Which of the ions given is the result of an alpha cleavage? (A) 29 (B) 31 (C) 43 (D) 56 (E) 70
A: the solution is shown below.
Q: These are particles that ionize in a solution. O a. nonelectrolytes O b. electrolytes O c. electrone...
A: Given, a. non-electrolytes b. electrolytes c. electronegativity d. electron Required, ...
Q: At a certain temperature the rate of this reaction is second order in N,0, with a rate constant of 9...
A: 2nd order reaction: 2N2O5(g) ------> 2N2O4(g) + O2(g) inital conc. = 0.24M k = 90.8M-1s-1
Q: 1. Dynamite 2. Table Salt 3. Water 4. Carbon Dioxide_ 5. Baking Soda
A: To write the formulas for all compounds, check the compounds present in it and write their formulas ...
Q: Provide the IUPAC name of the compound. Br
A: This question belong to nomenclature of organic compounds. In this case ring are main carbon chain a...
Q: Explain how you could separate an equimolar mixture of Compound X, Y and Z. w chart). он Compound X ...
A: This problem needs a practical knowledge. It is a practical thing to separate them.
Q: How much more heat is released from the combustion of one mole of octane (C8H18) than from the combu...
A: Combustion of hydrocarbon produces CO2 and H2O Heat released from the combustion is calculated on th...
Q: [2] A 20.0 mg sample of a protein is dissolved in water to make 25.0 ml of solution. The osmotic pre...
A: Given: Mass of protein = 20.0 mg = 0.020 g. (Since 1 g = 1000 ...
Q: Compare the-chemical reactivity of calcium and magnesium. Explain the red litmus paper result. Can y...
A:
Q: Direction: Complete the table by writing all types of intermolecular forces (IMF) that will exhibit ...
A: Hydrogen bonding can be formed between a hydrogen atom and F, N and O atoms.
Q: Select the arrangement in which these ionic substances are arranged from higher to lower potential e...
A: Lattice energy is a type of potential energy. Lattice energy ∝ z+z- -----(1) Lattice energy ∝ 1...
Q: 45 100 MS-NU-3166 80- 60 40 20 55 83 29 98 101 116 10 20 30 40 50 60 70 80 90 100 110 m/z Relative I...
A: Alpha-cleavage (α-cleavage) in organic chemistry refers to the act of breaking the carbon-carbon bon...
Q: Please create the titration curve of aspartate and annotate the corresponding isoforms.
A: Amino acids are amphoteric in nature due to the presence their ionizable α-amino and α-carboxylic ac...
Q: What are the steps in diluting high concentration of stock solutions into lower concentration standa...
A: During dilution, concentration changes but no of moles remain same. So, M1V1 = M2V2 Let- Concentrati...
Q: Describe a step-by-step procedure on how you would solve this problem. 25. The tabulated data show t...
A: The order of the reaction is calculated by hit and trial method
Q: 1. What produces the dry sensation in your mouth when drinking wine? 2. What happens when tannins co...
A: Human saliva is full of protein this makes it so slippery. When we drink wine the tannins and sa...
Q: What is the test to identify the haloalkane functional group? explain
A:
Q: If 1 pound = 2.205kg, how many grams is in 4.95 pounds? Report your answers to 2 decimal places with...
A:
Q: Draw the structure 3,5-diethyl-3-methyloctane in the window below.
A:
Q: - What is the ideal solvent behavior for crystallization? - You will probably hear me say frequen...
A: Crystallization is defined as the process in which atoms in a given sample are arranged into a highl...
Q: The structure given is Escitalopram, an active enantiomer of citalopram and works as an anti-depress...
A: Hybridisation can be defined as the process of mixing of pure atomic orbitals to form molecular orbi...
Q: raw the expected major product for both compounds A and B of the following reaction uence HCI A
A: Major product is analyse by applying appropriate reaction mechanism on substrate.
Q: Determine the mass in grams of HCI that can react with 0.750 g of AI(OH)s according to the following...
A:
Q: If an atom has 7 protons, how many electrons does it want to gain in order to fill its valence shell...
A:
Q: Why is malonic acid a diprotic acid? Please explain using the two ionization equations using chemica...
A: Molecular formula of malonic acid is HOOCCH2COOH
Q: The boiling point of an alkane is lower than the alkane immediately after it in the series. Select t...
A: Boiling point is the temperature at which a liquid converts into vapour at given pressure.
Q: Match each weak acid with the pH value at which it would buffer.
A: Weak acids are the acids that do not dissociate completely in aqueous solution. There exists an equi...
Q: Question 24 Rainfall does not contribute to a soil's acidity. A True B False Question 25 Molecules a...
A: Thus given statement is true that is acidic.
Q: Give the name for the following complexes according to the latest IUPA nomenclature. (i). [Sn(CN)3CI...
A: We have been given some coordination compounds and asked to do nomenclature.
Q: How many grams of nitrogen gas are present in a 10.0 L vessel at 19°C and 1.60 atm?
A:
Q: how how the forward and reverse reaction rates respond to having some HOCl suddenly removed from the...
A: The reaction rate depends on the concentration of the reactant but the reaction rate does not depend...
Q: Identify the hydrocarbon that gives the IR spectrum shown in Figure 14.50 and has an m/z 102 in its ...
A: Solution -
Q: Aqueous hydrochloric acid (HC1) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium ...
A:
Q: Chemistry Demonstrate 2 practical examples electrocoagulation, including redox half reactions.
A:
Q: determine the number of atoms of 23.64g of Ce
A: Given, mass of Ce = 23.64 g Molar mass of Ce = 140 g.mol-1
Q: Reset Help `N R–NH2 R^Br R–OH R Highest priority Lowest priority The correct ranking cannot be deter...
A: Here we have to write the decreasing order of priorities of the following groups according to CIP r...
Q: 2NO(g) + → N,O2(g) FAST N,0,(g) + 0,(g) → 2NO,(g) SLOW Determine the overall reaction O 2NO(g) + O2(...
A:
Q: 2. Explain each general trend that affects the Boiling point: A. Impurities B. Intermolecular force|
A: Boiling point can be defined as the temperature at which a liquid substance starts converting into v...
Q: If the purity of the naphthalene sample was 96.0%, what is the relative error of the experiment? Cru...
A:
Q: Calculate the [H+] in an HClO2 solution that has a pH of 2.59. (Answer to 4 decimal places
A:
Q: Rank the gasses from highest to lowest global warming potential. (1 being the highest, 4 being the l...
A: Due to global warming, the temperature of our earth increases day by day.

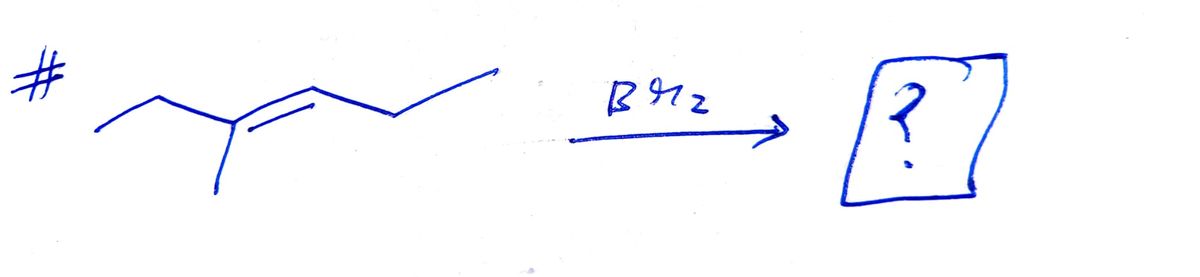
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- 2. Complete the following: Stereochemistry and regiochemistry may be important. type a) NaOH (aq) b) neutralize ➤ OMe a) LiAlH4, ether NMe2 f) b) H3O+ type b) a) HS SH ZnCl2, H+ b) Raney Ni g) type formation of thioacetal/desulfurization HO type Hell-Volhard-Zelinsky Reaction h) CO₂Et type Wittig Reaction H H₂N OEt OH DCC, CH2Cl2 Ph type a) NaH, THF CO₂Et d) b) BnBr, THE CO₂Et c) NaOH(aq); HCl(aq); heat type type HO type (COCI)2, cat DMF CH2Cl2 j) CI type a) NHEN b) LiAlH, THE c) H3O+ CO₂Me CO₂Me[Review Topics) [References] Draw the major product(s) of the following reactions including stereochemistry when it is appropriate. (CH)CHCH,-CEC-H 1 Br2 • Consider E/Zz stereochemistry of alkenes. • If there is more than one major product possible, draw all of them. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate multiple products using the + sign from the drop-down menu.see these instructions. Consider stereochemistry and enter either enantiomer (but not both) if a racemic mixture of products forms. If a mixture of two diastereomers is expected, enter either (but not both). 1. MgCl 2. H₂O
- up an example (not appearing in this ChemActivity) of a pair of molecules that are a)constitutional isomers, b) conformers. c) configurational stereoisomers.How many and what type of products will form if the following molecule undergoes E1? H Br choose your answer... V total product(s) form. choose your answer... V will be optically active.Fill in the boxes in the problem below to complete the reaction. STEREOCHEMISTRY EXISTS IN EACH OF THESE AND ALL POTENTIAL STEREOISOMERS THAT CAN RESULT/REACT SHOULD BE SHOWN. Et 1. BH, 2. H,O,, NAOH H,O H2 Pd For this last reaction with H2/Pd. (a) circle any chiral products. (b) Is the product mixture optically active? Yes or No (circle one!)
- [Review Topics] Either the reactant (X) or the major organic product is missing from the equation below. Draw the missing compound. [References) X + H2 H3C CH3 Pd/C CH,CHCHCHCH, • You do not have to consider stereochemistry. • In the case of a missing reactant, there may be more than one answer. If so, draw all possible reactants in separate windows. • Separate structures with + signs from the drop-down menu. (Previous Next ChemDoodle Save and Exit O 29 étv ll MacBook Air DII DD F11 F10 F9 80 888 FB F7 F5 F4 F3 * 一 & de %3D $ 8 9 7 4 { Y R H J K F G D ? + I" .. .-[Review Topics] [References) Either the reactant (X) or the major organic product is missing from the equation below. Draw the missing compound. CH3 Pd/C + H2 CH;CCH2CH3 CH3 • You do not have to consider stereochemistry. • In the case of a missing reactant, there may be more than one answer. If so, draw all possible reactants in separate windows. • Separate structures with + signs from the drop-down menu. opy aste ChemDoodle Previous NextI am studying so much but I am not sure if these are correct. Can you go over it plsss? A Hoffmann product is O the result of the fastest mechanistic process. the most highly substituted alkene possible. the same as the Zaitsev's product, but the term "Hoffmann" is used for E2. the most stable alkene. What does Zaitsev's rule state? As the degree of substitution around the C=C of an alkene decreases, the stability of alkene increases. None of the statements is correct. O As the degree of substitution around the C=C of an alkene decreases, the stability of alkene decreases. As the degree of substitution around the C=C of an alkene increases, the stability of alkene decreases.
- [References] Use the References to access important values if needed for this question. Draw the structure of the organic product that is expected when the following compound is treated with concentrated H,SO4. CH3 CH2CCH2CH3 tosH heat HO. • You do not have to consider stereochemistry. • In cases where there is more than one answer, just draw one. ed P. opy aste C. CH4 ChemDoodle Retry Entire Group 3 more group attempts remaining Submit Answer Previous Next Email Instructor Save andProvide the major product of the reaction sequence. If cis/trans isomers are possible, draw only the major isomer. If enantiomers are possible, do not specify configuration.1. Draw one of the two enantiomers of the major product from this 2. Draw the product of this reaction. Ignore inorganic byproducts. 3. Draw the starting structure that would lead to this major produ reaction. Use wedge and dash bonds to indicate (and its enantiomer) under these conditions. stereochemistry where appropriate. Ignore inorganic byproducts. 1. BH3-THF 2. H2O2, NaOH 1. BH3-THF 2. H2O2, NaOH 1. BH3-THF 2. H2O2, NaOH HO

