
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Draw a curved arrow mechanism of the following SN2' reaction. Make sure you show the intermediate structure.
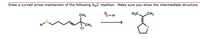
Transcribed Image Text:Draw a curved arrow mechanism of the following Sy2' reaction. Make sure you show the intermediate structure.
CH3
8-H
H3C,
CH3
CH3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- To preview the image Click Here Give a curved arrow-pushing mechanism for the following reaction and make sure to include all nonbonding electrons. CLEARLY Indicate the initiation and propagation steps. H-Br CH3OOCH3 :Br: Karrow_forward3. The haloalkane below is heated in a solution of ethanol and undergoes SN1 and E1S a) Draw the first step of the mechanism of the reaction. b) Show all the more stable carbocations that could be formed from (a). c) Draw all the El products that can be formed from (b). d) Circle the most favorable product(s) and explain your reasoning. e) Name: the most favored product(s).(Note: for cyclic alkenes the carbons of the C-C must be numbered 1,2 and in the name there is no need to specify its position). Brarrow_forwardSynthesis using a multiple RXN. A desired alkene product is shown below. A starting material is given. Propose a synthesis (2-3 step) of the molecule, include necessary reagents and the complete arrow. pushing mechanism for each reaction involved in the synthesis. S starting material Final productarrow_forward
- need answer in step by steparrow_forward1. For the substitution (SN2) reaction below circle the nucleophile and draw a box around the electrophile. Then, using curved arrows, draw the reaction mechanism. CH;S acetone Brarrow_forward(in first pic) need help with the last one, with the alkyne & the question in the 2nd pic. thank you!!arrow_forward
- An SN2 mechanism's top requirement from an aprotic solvent is to ensure that it can stabilize the leaving group.arrow_forward2. Draw the major product of the following E2 reaction. Show stereochemistry where appropriate. NaOCH2CH3 CH3CH2OH Next, draw the chair conformation and curved arrow mechanism that lead to your product you have provided.arrow_forwardDraw the product of the E2 reaction shown below. Include the correct stereochemistry. Use a dash or wedge bond to indicate relative stereochemistry on asymmetric centers, where applicable. Ignore any inorganic byproducts. CI Strong Basearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY