
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
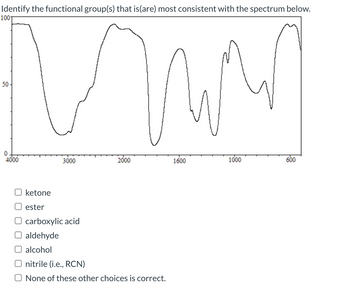
Transcribed Image Text:Identify the functional group(s) that is(are) most consistent with the spectrum below.
1001
www
1600
50-
0
4000
ketone
ester
3000
2000
carboxylic acid
aldehyde
alcohol
nitrile (i.e., RCN)
None of these other choices is correct.
1000
600
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Infrared Spectrum T (the left one) represents which of the following compounds? Notice the labels i and is on the two spectra close to the y-axis (transmittance) FIGURE 1 0 cyclohexanone 3000 INFRARED SPECTRUM phenylacetylene 2000 -COOH benzoic acid 1000 08 ii mm 02 3000 -OH phenol INFRARED SPECTRUM 2000 1000arrow_forwardAn uknown organic compound with molecular formula C9H10O2 find DUI, draw the structure of the compound on both HNMR and 13CNMR spectra and assign all peaks, please see attached picture belowarrow_forwardvalues : Experimental values of IR Peaks compared to literature values for Benzoic Acid (ATR method) Bond Stretch Functional group Experimental Peak(cm-1) Literature Peak(cm-1) O-H Carboxylic acid 2800 3200-2200 C=O Carboxylic Acid 1674.9 1677.5 C=C (ring) Aromatic stretch 1579.5 1581-1418.5 C-H Aromatic Sp2 3100 C-O stretch 1285.5 O-H Alcohol 929.89 table 3: Experimental values of IR peaks compared to literature values for 2-Naphthol Bond Stretch Functional group Experimental Peak(cm-1) Literature Peak(cm-1) O-H Alcohol 3219.1 3400-3080 C-C Aromatic (ring) stretch 1507.6 C=C Aromatic stretch 1597.7 1627.3-1377.8 C-H Aromatic Sp2 3050 C-O Secondary alcohol stretch 1168.9 IR results used to prove that the compounds are effectively separated. Discuss on bothdiagnostic peaks and fingerprint region.arrow_forward
- The acids and bases used in the first part of this experiment were strong acids and strong bases. What is a weak acid or base? simple plzarrow_forwardBoth of the mass spectra are of an aromatic compund. Identify the fragments.arrow_forwardAssign molecules B, C, D and F to the correct mass spectrum and provide the structure of thebase peak. draw the assigned moleculesarrow_forward
- In the following FTIR Spectrum, what functional group can you recognize? 100 Carboxylic acid Alcohol Ether O Alkene 3000 2000 www HAVENUMBERI-l 1500 1000 500arrow_forwardIn the attached H1-NMR, draw and identify each set of equivalent hydrogens in the structure and list peak they belong to in the NMR spectrum.arrow_forward8) In mass spectrometry (M.S.), the molecular ion (M) is usually the largest peak. For what type of compounds would you expect the (M-1)* peak be quite large. a) alkyl bromides c) alcohols e) alkyl chlorides b) aromatic compounds d) hydrocarbonsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY