
Human Anatomy & Physiology (11th Edition)
11th Edition
ISBN: 9780134580999
Author: Elaine N. Marieb, Katja N. Hoehn
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Dehydration synthesis always results in the formation of which of the following?
A.)Water
B.)Amino acids
C.)Proteins
D.)Peptides
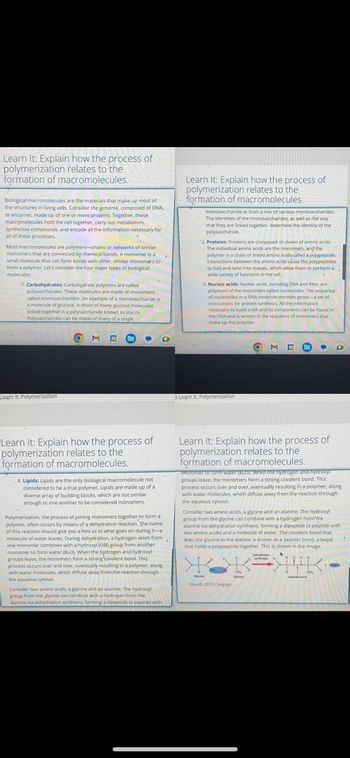
Transcribed Image Text:Learn It: Explain how the process of
polymerization relates to the
formation of macromolecules.
Biological macromolecules are the materials that make up most of
the structures in living cells. Consider the genome, composed of DNA,
or enzymes, made up of one or more proteins. Together, these
macromolecules hold the cell together, carry out metabolism,
synthesize compounds, and encode all the information necessary for
all of these processes.
Most macromolecules are polymers-chains or networks of similar
monomers that are connected by chemical bonds. A monomer is a
small molecule that can form bonds with other, similar monomers to
form a polymer. Let's consider the four major types of biological
molecules:
1. Carbohydrates: Carbohydrate polymers are called
polysaccharides. These molecules are made of monomers
called monosaccharides. An example of a monosaccharide is
a molecule of glucose. A chain of many glucose molecules
linked together is a polysaccharide known as starch.
Polysaccharides can be made of many of a single
Learn It: Polymerization
O M D
- C
Learn It: Explain how the process of
polymerization relates to the
formation of macromolecules.
4. Lipids: Lipids are the only biological macromolecule not
considered to be a true polymer. Lipids are made up of a
diverse array of building blocks, which are not similar.
enough to one another to be considered monomers
Polymerization, the process of joining monomers together to form a
polymer, often occurs by means of a dehydration reaction. The name
of this reaction should give you a hint as to what goes on during it-a
molecule of water leaves. During dehydration, a hydrogen atom from
one monomer combines with a hydroxyl (OH) group from another
monomer to form water (H₂O). When the hydrogen and hydroxyl
groups leave, the monomers form a strong covalent bond. This
process occurs over and over, eventually resulting in a polymer, along
with water molecules, which diffuse away from the reaction through
the aqueous cytosol.
Consider two amino acids, a glycine and an alanine. The hydroxyl
group from the glycine can combine with a hydrogen from the
ya
alanine via dehydration synthesis, forming a dipeptide (a peptide with
.
Learn It: Explain how the process of
polymerization relates to the
formation of macromolecules.
monosaccharide or from a mix of various monosaccharides.
The iden
identities of the monosaccharides, as well as the way
that they are linked together, determine the identity of the
polysaccharide.
2. Proteins: Proteins are composed of chains of amino acids.
The individual amino acids are the monomers, and the
polymer is a chain of linked amino acids called a polypeptide.
Interactions between the amino acids cause the polypeptides
to fold and twist into shapes, which allow them to perform a
wide variety of functions in the cell.
3. Nucleic acids: Nucleic acids, including DNA and RNA, are
polymers of the monomers called nucleotides. The sequence
of nucleotides in a DNA molecule encodes genes-a set of
instructions for protein synthesis. All the information
necessary to build a cell and its components can be found in
the DNA and is written in the sequence of monomers that
make up this polymer.
3 Learn It: Polymerization
Learn It: Explain how the process of
polymerization relates to the
formation of macromolecules.
monomer to form water (BO). Wisen the hydrogen and hydroxyl
groups leave, the monomers form a strong covalent bond. This
process occurs over and over, eventually resulting in a polymer, along.
with water molecules, which diffuse away from the reaction through
the aqueous cytosol.
MD O - C
Consider two amino acids, a glycine and an alanine. The hydroxyl
group from the glycine can combine with a hydrogen from the
alanine via dehydration synthesis, forming a dipeptide (a peptide with
two amino acids) and a molecule of water. The covalent bond that
links the glycine to the alanine is known as a peptide bond, a bond
that holds a polypeptide together. This is shown in the image.
Gine
† Cengage
CH₂
Na
Dehydration
ap
><
CH₂
peptide bond
95
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Carbons is used in cells for a) forming the charge across the membrane by serving a positive cations b) as the structural backbone of most molecules c) forming the second most common element D) none of thesearrow_forward1. Indicates whether the statement is true (T) or false (F). If the statement is wrong, rephrase it so that it is true. a) Glycogen is a lipid b) Passive transport moves substances against a concentration gradient. c) Starch is a disaccharide. d) Facilitated diffusion is an example of passive transport.arrow_forward3. Place the following in correct sequence from largest to smallest: a) molecule b) electron c) cell d) organ e) atom f) proton g) tissuearrow_forward
- DNA is composed of which are attached to each other by bonds. O A) nucleotides. phosphodiester O B) amino acids.. peptide C) monosaccharides. glycosidic O D) nucleotides. hydrogen O E) amino acids.. hydrogenarrow_forwardWhich of the following is an example of secondary protein structure?A) DipeptideB) TriglycerideC)α -helixD) Amino acidE) Fatty acidarrow_forwardWhat is the name of the bond formed between adjacent amino acids in a protein?Question 34 options: A) hydrogen B) carboxyl C) peptide D) ester E) phosphodiesterarrow_forward
- What reagent present in the protein sample buffer is important for breaking disulfide bonds? a) 2-mercaptoethanol O b) chloride ions O c) glycine O d) SDSarrow_forwardThe movement of water along a concentration gradient through a semi-permeable membrane: A) osmosis B) endocytosis C) diffusion D) active transport Question 4 Which bonds are created when the primary structure of a protein forms? OA) peptide bonds B) ionic bonds C) glycosidic bonds D) hydrogen bondsarrow_forwardWhich of the following describes a primary protein structure?A) Protein structure maintained by disulfide linkages.B) Amino acid sequence maintained by peptide bonds.C) Protein chains maintained by interactions of peptide backbones like an α-helix.D) Arrangement of multiple protein subunits.E) Protein structure maintained through multiple hydrogen bonds.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education