
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
give explanation part in typed form and diagram in clean paper
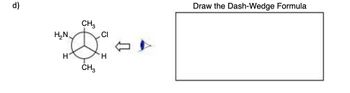
Transcribed Image Text:6
H₂N
H
CH3
CH3
CI
H
Draw the Dash-Wedge Formula
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine the mass of oxygen in a 6.8-g sample of Al2(SO4)3. Express your answer to two significant figures and include the appropriate units.arrow_forwardCHRIRAD mework WP NWP Assessment Player Ul Appli X tion.wiley.com/was/ui/v2/assessment-player/index.html?launchld=d7fbf299-2b78-4aab-9fe4-25d65ce73435#/question/5 Question 6 of 6 W Carbon tetrachloride (CC14) was prepared by reacting 119 g of carbon disulfide and 119 g of chlorine. Calculate the percent yield if 65.0 g of CCl4 were obtained from the reaction CS₂+3 Cl₂ → CCl4 + S₂Cl2. Save for Later + % yield Want to see a relevant text example? GO Tutorial -/2 = O Search کا Attempts: 0 of 5 used Submit Answerarrow_forwardOnly typed solutionarrow_forward
- Referring to your best straight line graph, what are the units of the slope?What physical property of steel is the slope of this line related to? Use the graphed data and your best trendline equation to predict the diameters of the three unknown ball bearings.Show your calculations below and report your three results in the data table above with 2 decimal place accuracy. ] 7. How could we improve the accuracy of the largest unknown ball bearing's diameter prediction? can you help me answer all this questions please, ai really appreciated it.arrow_forwardI need help finding the volume of stock and water given thetarget concentration. I dont know how to use the diluation equation to find it with the data provided. I need step by step instructions on how to get them, then for the conductivity measurements, how do i find concentration (m) with the given infoarrow_forwardSelect all true statements. ANFO explosive ...1-is more sensitive when density is higher than 1.2g/cm32-produces carbon monoxide (CO) when in a fuel shortage3-has a low velocity of detonation (VOD) and is useful to produce heave action forsoft and fractured rocks4-should not be stored for long periods of time5-has a higher velocity of detonation (VOD) when used in small diameter blast holes6-can be over-oiled (fuel excess) to account for potential fuel evaporation duringhot summer monthsarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY