
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
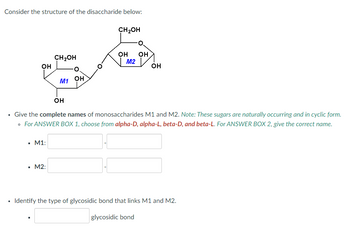
Transcribed Image Text:Consider the structure of the disaccharide below:
OH
I ▪ M1:
CH₂OH
I M2:
M1
OH
CH₂OH
OH
M2
OH
OH
Give the complete names of monosaccharides M1 and M2. Note: These sugars are naturally occurring and in cyclic form.
• For ANSWER BOX 1, choose from alpha-D, alpha-L, beta-D, and beta-L. For ANSWER BOX 2, give the correct name.
OH
Identify the type of glycosidic bond that links M1 and M2.
glycosidic bond
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A monosaccharide that consists of 4 carbon atoms, one of which is part of a aldehyde functional group, is classified as a(n) a). Aldotetrose b). Aldohexose c). Ketotetrose d). Ketohexosearrow_forwarda. State the type of glycosidic bond that is present in the disaccharide shown below. b. Is the disaccharide shown below a reducing sugar (Yes or No)? Explain why writing complete sentences. CH2OH CH2OH H H H H OH H ОН Н ОН Н НО H H OH H ОНarrow_forwardDraw the Haworth projection of the disaccharide made by joining two D-glucose molecules with an a(1→6) glycosidic bond. If the disaccharide has more than one anomer, you can draw any of them. Click and drag to start drawing a structure. × Śarrow_forward
- Describe the fate of soluble and insoluble, inorganic and organic fractions as they areprocessed through different unit operations during conventional wastewatertreatment.arrow_forward5. Draw the structure of a disaccharide that consists of two a-galactoses connected by an al-4 linkage.arrow_forwardFor the disaccharide, give the "numbers" for the glycoside bond (reading left to right). HO OH OH OH OH OHarrow_forward
- Draw the structure of a disaccharide made up of two D-glucose units. The glycosidic bond is α-1,6-glycosidic bond.arrow_forwardWhich monosaccharides form: a) lactose b) maltose c) sucrosearrow_forwardif the disaccharide maltose is formed from two glucose monosaccharides which are hexose sugars how many atoms of carbon hydrogen and oygen does maltose contain and why?arrow_forward
- Consider N-acetyl-d-glucosamine Q.) Draw a chair conformation for the disaccharide formed by joining two units of the pyranose form of N-acetyl-d-glucosamine by a b-1,4-glycosidic bond. If you draw this correctly, you have the structural formula for the repeating dimer of chitin, the structural polysaccharide component of the shell of lobsters and other crustaceans.arrow_forwardExplain the Cyclic Forms of Monosaccharides ?arrow_forwardClick the "draw structure" button to launch the drawing utility. Consider the following monosaccharide. H draw structure ... HO- -C-H HO–C—H HO- -C-H H-C-OH CH₂OH Draw the a anomer of the cyclic form.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY