
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question

Transcribed Image Text:Consider the following equilibrium at 416.2 K:
NH4HS(s) NH3(g) + H₂S(g)
The partial pressure of each gas is 0.102 atm. Calculate Kp and Ke for the reaction.
Enter your answers in scientific notation.
Kp=
Kc
=
x 10
x 10
Expert Solution
arrow_forward
Step 1
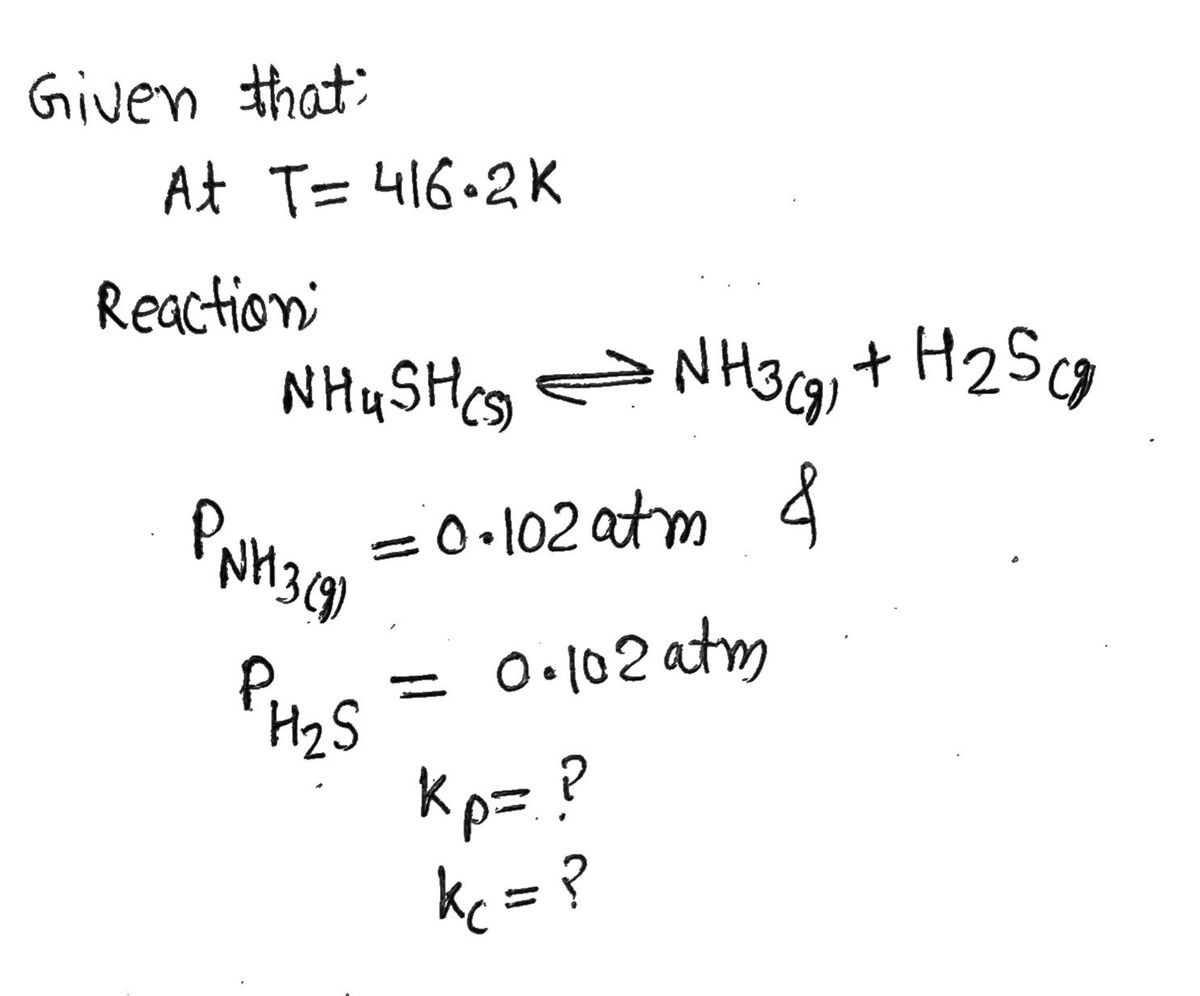
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- VIII. Exercise 2.2 Write balanced equations for the following Redox reactions: 1. P4(s) + Cl(aq) → PH3(g) + Cl2(g) 2. MnO4 (aq) + NO₂ (aq) → Mn²+ (aq) + NO3 (aq) in acid solution 3. C₂H4 (g) + BiO₂ → CO2(g) + Bi³+ in basic solution (aq) 4. PbO2 (s) + H₂O → O2(g) + Pb²+ 5. CrO4 (aq) (aq) 2- + SO3 → Cr³+ SO4²- (aq) (aq) (aq) in acid solution in basic solution in acid solutionarrow_forwardWhen 1.00 mol of H2O and 1.00 mol of CO are introduced into an empty 1.00 L container at 960 K and allowed to come to equilibrium, the mixture contains 0.422 mol of water vapor. Write the equilibrium expression for the reaction and calculate the value of Kcarrow_forwardConsider the following chemical equilibrium: C(s) + 2 H₂ (g) ⇒ CH₂(g) Now write an equation below that shows how to calculate K from K for this reaction at an absolute temperature T. You can assume I is comfortably above room temperature. If you include any common physical constants in your equation be sure you use their standard symbols, found in the ALEKS Calculator. K -0 X 00arrow_forward
- Consider the equilibrium system described by the chemical reaction below. If the partial pressures at equilibrium of NO, Cl2, and NOCI are 0.095 atm, 0.171 atm, and 0.28 atm, respectively, in a reaction vessel of 7.00 L at 500 K, what is the value of Kp for this reaction? 2 NO(g) + Cl2(g) = 2 NOCI(g)arrow_forwardConsider the following equilibrium: N2O4(g) 2NO2 (g) AG = 5.4 kJ Now suppose a reaction vessel is filled with 2.78 atm of dinitrogen tetroxide (N204) at 710. °C. Answer the following questions about this system: rise x10 fall Under these conditions, will the pressure of N2O4 tend to rise or fall? Is it possible to reverse this tendency by adding NO2? In other words, if you said the pressure of N2O4 will tend to rise, can that be changed to a tendency to fall by adding NO2? Similarly, if you said the pressure of N2O4 will tend to fall, can that be changed to a tendency to rise by adding NO2? If you said the tendency can be reversed in the second question, calculate the minimum pressure of NO2 needed to reverse it. Round your answer to 2 significant digits. yes no ☐ atm X Sarrow_forwardConsider the following equilibrium: 2NOC1 (g) → 2NO(g) + Cl₂ (g) AG = 41. kJ Now suppose a reaction vessel is filled with 8.30 atm of nitrosyl chloride (NOC1) and 5.80 atm of chlorine (C1₂) at 878. °C. Answer the following questions about this system: Under these conditions, will the pressure of Cl₂ tend to rise or fall? Is it possible to reverse this tendency by adding NO? In other words, if you said the pressure of Cl₂ will tend to rise, can that be changed to a tendency to fall by adding NO? Similarly, if you said the pressure of Cl₂ will tend to fall, can that be changed to a tendency to rise by adding NO? If you said the tendency can be reversed in the second question, calculate the minimum pressure of NO needed to reverse it. Round your answer to 2 significant digits. оо rise fall yes no 0 atm x10 X Śarrow_forward
- 3. Phosphoryl chloride, POCI3(g), is used in the manufacturing of flame retardants. It is manufactured in an equilibrium process in which phosphorus trichloride reacts with nitrogen dioxide to form POCI3(8)and NO(g) according to the following equation: PCI3 (g) + NO2 (g) = POCI3 (g) + NO (g) The Keg for this reaction at 800 K is 6.90. If 1.25 mol PCI3 (g) and 1.94 mol NO2 (g) are placed into a 2.0 L container, what is the concentration of each gas at equilibrium at 800 K? PCI3 (g) NO2 (g) POCI3 (g) NO(g) Keq = 6.90 + + Earrow_forwardConsider the following equilibrium: 2NH3(g) = N₂ (g) + 3H₂(g) 2 AG = 34. kJ Now suppose a reaction vessel is filled with 7.87 atm of ammonia (NH3) and 1.17 atm of hydrogen (H₂) at 448. °C. Answer the following questions about this system: Under these conditions, will the pressure of NH3 tend to rise or fall? Is it possible to reverse this tendency by adding N₂? In other words, if you said the pressure of NH3 will tend to rise, can that be changed to a tendency to fall by adding N₂? Similarly, if you said the pressure of NH3 will tend to fall, can that be changed to a tendency to rise by adding N₂? If you said the tendency can be reversed in the second question, calculate the minimum pressure of N₂ needed to reverse it. Round your answer to 2 significant digits. rise fall yes no atm x10 X 5arrow_forwardUsing any data you can find in the ALEKS Data resource, calculate the equilibrium constant K at 25.0 °C for the following reaction. 2 NO (g) N2(g) + O2(g) Round your answer to 2 significant digits. K ☐ x10 คarrow_forward
- An empty steel container is filled with 3.10 atm of H₂ and 3.10 atm of F2. The system is allowed to reach equilibrium. If Kp = 0.450 for the reaction below, what is the equilibrium partial pressure of HF? H₂ (g) + F₂ (g) → 2 HF (g)arrow_forwardThe steam reforming reaction of methane follows this chemical equation: CHA(g) + H50(g) - CO(g) + 3 H2(g) A H° = +193 kJ/mol At 298 K the reaction lies far to the reactant side with a very small equilibrium constant of 2.35 × 10-23. To obtain a significant amount of the desired hydrogen gas product, the reaction is operated at very high temperatures. What is the equilibrium constant at 1100 K?arrow_forwardConsider the following equilibrium: 2NH3(g) → N₂(g) + 3H₂(g) AG = 34. kJ Now suppose a reaction vessel is filled with 6.39 atm of ammonia (NH3) and 0.200 atm of hydrogen (H₂) at 206. °C. Answer the following questions about this system: Under these conditions, will the pressure of NH3 tend to rise or fall? Is it possible to reverse this tendency by adding N₂? In other words, if you said the pressure of NH3 will tend to rise, can that be changed to a tendency to fall by adding N₂? Similarly, if you said the pressure of NH3 will tend to fall, can that be changed to a tendency to rise by adding N₂? If you said the tendency can be reversed in the second question, calculate the minimum pressure of N₂ needed to reverse it. Round your answer to 2 significant digits. OO U rise fall yes no atm x10 X Śarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY