Q: Safrole is used as a topical antiseptic. Calculate the vapor pressure of a solution prepared by diss...
A:
Q: 22. How many Pop TartsM are needed to convert 1,000.0 g of water at 20.0°C to 100.0°C? One Pop Tart™...
A: Given : Mass of water = 1000g
Q: Write a net ionic equation for the overall reaction that Occurs when aqueous solutions of phosphoric...
A: The reactants given are phosphoric acid i.e. H3PO4 and sodium hydroxide i.e. NaOH.
Q: 3. The density of an unknown liquid is 0.874g/mL. What is the mass of the liquid if a graduated cyli...
A:
Q: "green" oxidation The above reaction is one of example of environmental benign synthesis which does ...
A: Green synthesis is an environmentally benign synthesis which does not involve organic solvent and to...
Q: i. LIAIH4 i. deprotection CO2Me J CH2=CHCOCH3 protection ii. H3O* ii. dehydration K The synthesis of...
A:
Q: Match the colligative property is applied in the given situation. Blake wants to cook macaroni fast ...
A:
Q: 6. What is the product of the following reaction? CHO Ph;P- CH2 OH PPH3 II II IV А) I B) П С) II D) ...
A:
Q: (Q77) Select all the methods that may generally be used to speed up a reaction: O Grinding a chunky ...
A:
Q: Question 3 Modified True or False. Caffeine is a natural stimulant most commonly found in tea, coffe...
A: Caffeine is a natural stimulant. It is found in black and green tea, cocoa and coffee plants.
Q: (Q79) Using the information provided, what are the reaction orders for CHCI3 and Cl2 respectively? C...
A:
Q: In a gas mixture, the partial pressures are argon 440 mmHg , neon 85 mmHg , and nitrogen 130 mmHg.Wh...
A: Given :- Partial pressure of argon = 440 mm Hg Partial pressure of neon = 85 mm Hg Partial pressu...
Q: A 66.7 mL sample of 18.0 M sulfuric acid was diluted with enough water in a volumetric flask to make...
A: The solution is as follows:
Q: Propose a reasonable structure based on the molecular formula, the H NMR, and the proton-decoupled 1...
A: Given, Formula of unknown compound = C7H16O
Q: Based on the following figure, explain the effect of stray radiation on absorption measurement. 0.0%...
A: A question based on concentration terms that is to be accomplished.
Q: Modified True or False. The amount of caffeine was influenced by the temperature of water used to br...
A:
Q: The equilibrium constant, Kp, for the following reaction is 3.11×10-3 at 350 K: 2IBr(g) Br2(g) + I2...
A:
Q: CH;-C-CH3 CH,CH2CH2-C-CH,CH2-C-CH3 ÇH,CH3 H H CH,CH2CH3 CH3-ÇHCH,-C-CH,
A: We are to write the IUPAC name of given structures. we will follow the longest chain and substituent...
Q: A gas mixture has a molar composition of 10% H2, 10% O2, 30% CO2 and balance H0. What is the molar c...
A: Given: The molar compositions of H2, O2 and CO2 are 10%, 10% and 30% respectively.
Q: Calculate the amount of heat needed to boil 80.0g of hexane (C6H14), beginning from a temperature of...
A:
Q: Based on the FTIR spectra, identify the functional group and its description based on the assigned w...
A: Given IR Spectrum:
Q: What is the volume, in liters, of 4.00 molesmoles of methane ga
A: From ideal gas equation P V = n R T where P = Pressure of the gas = 2 atm V = Volume of gas = ...
Q: For which of the following reactions does an increase in pressure have no effect on the equilibrium?...
A: For which of the following reaction : increase in pressure doesn't affect the equilibrium ? a) 2CO2...
Q: 7. Convert 24.5°F to degrees Celsius and Kelvin
A:
Q: Info on Oxalic acid is: pKa1= 1.25 pKa2= 3.81
A: Molarity of aqueous solution of H2C2O4 = 0.22 M
Q: Show the product, including stereochemistry, of the reaction of the epoxide below with Na+ -CN, H2O.
A: Three membered rings formed by two carbon and one oxygen atom is known as epoxide ring. The heterocy...
Q: C6H10O4 I need help answering 2 3 4 5 For IHD please show step by step ir analysis, frequency, ...
A: Qualitative analysis is a type of analysis that help in determining the identity or the components p...
Q: The solution from which a precipitate was formed. a. Mother liqour b. Titrant c. Saturated solution...
A: Mother liquor is defined as the solution which is left out after a certain component has been remove...
Q: - Part A If the initial cyclopropane concetration is 0.0420 M, what is the cyclopropane concentratio...
A:
Q: As the molar absorptivity increases, the concentration of caffeine_ A.Remain the same/not dependent ...
A: Since you have posted question with multiple subparts as per guidelines we can answer only three per...
Q: which two compounds are most similar in strength of their inter- or intramolecular forces? K3PO4, C...
A: To compare the strength of intermolecular interaction forces among two compounds , we have to know t...
Q: 2. Consider the reaction A+2B C whose rate at 25 °C was measured using three different sets of initi...
A: Equation A+2B <-> C
Q: Draw the simplest mechanism possible for the reaction below. You may need to re-draw structures to s...
A: Given : We have to draw the bond line structure.
Q: Pyrrole acylation Explain with mechanisms the formatiom of both products and also explain why these ...
A: We have to predict the mechanism of formation of products and reason for the proportion.
Q: Determine the enthalpy of the reaction: 3Fe,O3 (s) + CO (g)→ CO2 (g) + 2Fe;O4 (s), given the followi...
A:
Q: How many milliliters of 0.110 MHNO3 contain 7.68 g HNO3?
A: Given :- molar concentration of HNO3 solution = 0.110 mol/L mass of HNO3 = 7.68 g To calculate :- ...
Q: Describe a solution. What kind of mixture is considered a solution: homo- or heterogeneous, explain ...
A: Mixtures are defined as those substances or materials which are made of two or more components (mate...
Q: Mercury compounds are very poisonous. Mercury(II) nitrate reacts with sodium sulfide to form mercury...
A:
Q: A 0.239 g sample of carbon dioxide, CO2, has a volume of 555 mLmL and a pressure of 488 mmHg .
A: Given, Mass of carbon dioxide (CO2) = 0.239 g Volume (V) = 555 mL Pressure (P) = 488 mmHg Temperatu...
Q: The air in a 4.00 LL tank has a pressure of 1.90 atmatm . What is the final pressure, in atmospheres...
A:
Q: when aqueous solutions of potassium hydroxide and hydroiodic acid are combined. (Use Ht instead of H...
A:
Q: How many moles of C,H, contain 4.96 x 10 24 hydrogen atoms?
A: The number of moles of C3H8 required can be calculated by using the unitary method. C3H8 (propane) c...
Q: Rank the following compounds in order of decreasing acidity. Start with the most acidic compound. CN...
A:
Q: For the reaction given below, the value of the equilibrium constant at a certain temperature is 1.80...
A:
Q: The rate of a certain reaction is given by the following rate law: rate =k[NO]°[0,] Use this informa...
A: Chemical kinetics can be defined as the branch of chemistry that deals with rates of chemical reacti...
Q: this question. Be sure to specify states such as (aq) or (s). If a box is not needed leave it blank....
A:
Q: CIO4 + Pb+ OH" CIO3 + HPBO2 In the above redox reaction, use oxidation numbers to identify the eleme...
A: The element which loss electrons this is called oxidised ,and which gain electrons this is call redu...
Q: Question 3 Modified True or False. Caffeine is a natural stimulant most commonly found in tea, coffe...
A: Modified true or False is given below.
Q: What is the expression for the equilibrium constant for the reaction below? 2NH3(g) → N2(g) + 3H2(g)...
A:
Q: A solution is made by mixing 38.0 mL of ethanol, C,H,O, and 62.0 mL of water. Assuming ideal behavio...
A:

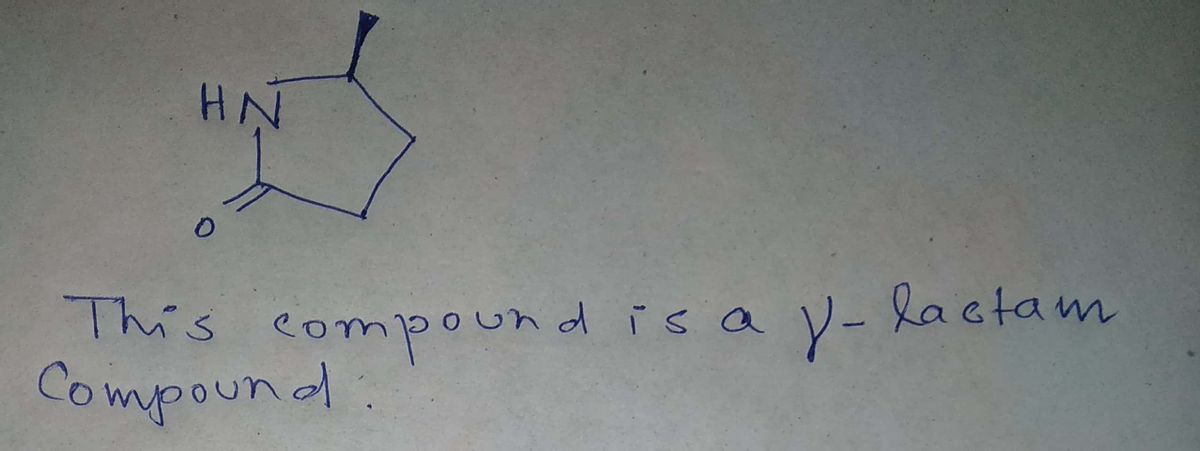
Step by step
Solved in 2 steps with 2 images


