Q: Identify each of the following compounds from its molecular formula and its 1H NMR spectrum:
A: 1H NMR- It is application of NMR spectroscopy called as proton Nuclear Magnetic resonance…
Q: For each molecule below, indicate how many signals you would expect in a 13C NMR. N' 4 6. 7
A:
Q: Identify each compound below from its molecular formula and its 13C NMR spectrum.
A: The 13C NMR always helps in recognizing carbon atoms that exist in given organic molecule. Each…
Q: Give the structure that corresponds to the following molecular formula and ¹H NMR spectrum::…
A: Secondary hydrogens are represented by the chemical shift (1.93); tertiary hydrogen carbon connected…
Q: Identify the following compounds from its molecular formula and its IR and 1H NMR spectra:
A: IR spectroscopy- is the vibrational motions of the atoms that are important. This theory utilizes…
Q: Identify each compound from its molecular formula and its 1H NMR spectrum: C9H12
A: To see the presence of double bond we calculate the degree of unsaturation for the given compound…
Q: What is the structure of compound X (molecular formula C3H&O) that gives the following proton NMR…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: (d) (9) OH OH Br CH3
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Compound P has molecular formula C5H9C102. Deduce the structure of P from its 1H and 13C NMR…
A: In NMR spectroscopy, splitting of signal takes place according to number of protons present on…
Q: Provide a structure for the given compound. C,H1,O2; IR: 1743 cm-l; 'H NMR spectrum chemical shift,…
A: Splitting pattern in 1H NMR is assigned according to n+1 rule where n represent the number of…
Q: Deduce the structure given the 1H-NMR and IR spectrum. Formula is C5H1002. Name structure if…
A:
Q: Identify compound below from its molecular formula and its 13C NMR spectrum.
A:
Q: C,H,NO, 2 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0 ppm
A: The NMR spectrum provides the following details about the structure of the molecules: The number of…
Q: 5. How might the two trimethylcyclohexane isomers shown below be most readily distinguished using…
A:
Q: An unknown compound X has the molecular formula C6H12O2 and the following H-NMR spectrum. What is…
A:
Q: Give the structure that corresponds to the following molecular formula and 'H NMR spectrum: C,H,F,I:…
A: The analysis of the results obtained from proton NMR, 13C-NMR, and mass spectra such as HRMS and…
Q: Which of the following compounds is responsible for the 1H NMR spectrum shown below?
A: Given spectrum shows 2 signals. One at 2.3ppm and another at 7.1ppm. The peak at 7.1ppm is the…
Q: How many peaks would you expect to see in the 13C-NMR spectrum of the molecule shown below? ㅅ 5 07 6…
A:
Q: Identify compound from its molecular formula and its 1H NMR spectrum:
A: First, calculate double bond equivalent of C9H12. It is given as: DBE = C + 1 - H2 - X2 + N2= 9 + 1…
Q: 8.What is the structure for the compound with a molecular formula CsH10Br2 with the following 13C…
A: Molecular formula = C5H10Br2
Q: Compound P has molecular formula C5H9ClO2. Deduce the structure of Pfrom its 1H and 13C NMR spectra.
A: Calculation of double bond equivalence: DBE = C +1 – (H + X – N) / 2 Here, C is the number of carbon…
Q: The H NMR, 13C NMR and mass spectra of an unknown compound provide the following information: Proton…
A:
Q: The 'H NMR, 13C NMR and mass spectra of an unknown compound provide the following information:…
A:
Q: Give the structure that corresponds to the following molecular formula and 'H NMR spectrum: C,H,F,I:…
A: The structure of the compound having molecular formula C2H2F3I is given below
Q: Identify compound from its molecular formula and its 1H NMR spectrum:
A: First, calculate the double bond equivalent as shown as follows: DBE = C + 1 - H2 - X 2 + N2= 9 + 1…
Q: The 1H NMR spectra of two compounds, each with molecular formula C11H16, are shown here. Identify…
A: (a)
Q: Give the structure that corresponds to the following molecular formula and 'H NMR spectrum: C,H12: 8…
A:
Q: (üi) H;C-ċ-CH2, CHT (c) How can the following compounds be distinguished. by . 'H-NMR? . Provide…
A:
Q: How many 13C-NMR signals does Toluene have? a) 2 b) 3 c) 4 d) 5
A: 13C-NMR (C-13 nuclear magnetic resonance) is a spectroscopic technique that is used to determine the…
Q: Identify each compound from its molecular formula and its 1H NMR spectrum: C9H12
A: To see the presence of double bond we calculate the degree of unsaturation for the given compound…
Q: Using the molecule below, how many signals are present in its 13C NMR & 1H NMR?
A:
Q: Analyze the following 'H-NMR spectrum; calculate DU, assign all the 'H-signals and give the…
A: NMR spectroscopy stands for Nuclear Magnetic Resonance Spectroscopy. It is also called proton-NMR…
Q: 3. How many signals would you expect in the 'H and 13C NMR spectrum of each molecule? H. ethyl…
A: The number of signals in 1H-NMR gives information about the number of different types of hydrogen…
Q: Identify each compound below from its molecular formula and its 13C NMR spectrum.
A: Calculation of double bond equivalent :
Q: Which structure of molecular formula, C7H140, fits the proton NMR spectrum shown below? doublet…
A: Given, The molecular formula of the compound C7H14O, the structure of the compound based on spectra…
Q: Identify compound below from its molecular formula and its 13C NMR spectrum.
A: 13C NMR spectroscopy studies the carbon-13 nucleus in the compound, and it provides the information…
Q: The 'H NMR and 13C spectra of a compound with a molecular formula of C,H1202 are shown below. 1.…
A: The structure of the compound is shown below:
Q: The 1H-NMR spectrum of Compound C shows five signals – δ 2.38 (1H, dt), 2.72 (1H, dt), 5.34 (1H, t),…
A: To identify the structure of the compound using NMR and mass spectra data.
Q: Identify compound below from its molecular formula and its 13C NMR spectrum.
A:
Q: Identify the following compounds from its molecular formula and its IR and 1H NMR spectra:
A: On the basis of all interpretation structure of compound is
Q: For the following compound, predict the number of signals in a 13C NMR spectrum: ∞ OA5 OB. 6 O C7 O…
A:
Q: How many carbon signals are expected in the proton-decoupled 13C NMR spectrum of the compound shown…
A:
Q: doublet 2H triplet 2H triplet 1H PPM NO2 NH2 NO2 NO2 NH2 A B D E
A: Ans B
Q: Q3/ B. Explain how you can identification the organic compound (C10H12O3) through the following…
A:
Q: Identify each of the following compounds from its molecular formula and its 1H NMR spectrum:
A: a) Calculation of the Double Bond Equivalence of C6H12O: where C is the no. of Carbon, H is the no.…
Q: Provide the correct structure for the compound with the following 1H NMR spectrum and molecular…
A: 1H NMR spectroscopy is used to determine the structure of compound with respect to hydrogen nuclei…
Q: Compound P has molecular formula C5H9ClO2. Deduce the structure of P from its 1H and 13C NMR…
A: Given molecular formula is C5H9ClO2. Observing the proton NMR gives the following information, Shift…
Q: 1. An unknown compound X has a molecular formula C13H19NO and produced a [M]+ ion at m/z 205. Its IR…
A: The degree of unsaturation or the number of multiple bonds, number of rings present in a molecule is…
Q: Propose a structural formula for compound J, molecular formula C,H,O, consistent with the following…
A: Given: Molecular formula =C3H6O To find: The structural formula of the compound J
Q: Identify each compound from its molecular formula and its 1H NMR spectrum: C9H12
A: The double bond equivalent for the compound is equal to 1. This suggests the presence of carbonyl…
Identify compound below from its molecular formula and its 13C NMR spectrum.

Calculation of double bond equivalent:
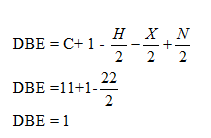
Step by step
Solved in 2 steps with 2 images

- Given C6H12O2 chemical formula. chemical shift , intergration, multiplicity and interpretation needed.Quantitative Analysis by External Standard Method Calcium in a juice sample is determined by atomic absorption spectrophotometry. A stock solution of calcium is prepared by dissolving 1.834 g of CaCl, 2H20 in water and diluting to 1000.0 mL. A substock calcium solution was prepared by transferring 10.00 mL of the stock to a 100.00 mL volumetric flask and diluting to the mark. Three standard solutions of calcium were prepared by transferring 2.500 mL. 5.00 mL and 10.00 mL of substock to 50.00 mL volumetric flasks and diluting to the mark. The sample is prepared by transferring 1.000 mL of fruit juice from the original bottle to a 25.00 mL flask and diluting to the mark. Strontium chloride is added to all solutions before dilution to avoid phosphate interference. Instrumental response of the standards is measured against the blank, and the following readings are recorded: 9.10, 18.6 and 37.0. The sampie reading is 28.1. Using the multipoint method, find the concentration of calcium in…In order to measure riboflavin in a breakfast cereal, a 5.00-gram sample of the cereal was extracted with acetic acid and filtered. The extract was transferred to a 50.0 mL volumetric flask and diluted to the mark. The sample was transferred to a cuvette and the fluorescence was measured. The instrument read 59 units (Fluorescence units –an arbitrary scale). 24.00 mL of the cereal extract was sample was mixed with 1.0 mL of a riboflavin standard that was 0.500 ug/mL. The fluorescence of this mixture was measured and read 94 fluorescence units. Calculate the concentration of riboflavin per gram of cereal (use µg/gram of cereal).
- 1-31. We shall learn when we study molecular spectroscopy that heteronuclear diatomic molecules absorb radiation in the microwave region and that one determines directly the so-called rotational constant of the molecule defined by B = 8721 where I is the moment of inertia of the molecule. Given that B = 3.13 × 105 MHz for H35C1, calculate the internuclear separation for H35C1. The atomic mass of H and 35C1 are 1.008 and 34.97, respectively.Ch-8-Cル-8-4s 2) ち* Maen %23 KMCA 写+ NooM.Chemistry 0.1237 (±0.0001) g of sodium carbonate (Na2CO3) required 22.46 (±0.02) cm3 of aqueous HCl for complete neutralisation. Determine the concentration of the HCl in mol dm–3 together with its uncertainty in mol dm–3 to an appropriate number of digits. You may assume the error in the molar mass of Na2CO3 is negligible.
- Staple your graph to the data sheet. Complete the following table: T-1(K-1) In(VP) 353 -0.00297 348 - o 06191 343 =0.00296 338 333=0.0300 323-0.0030u る= =0.00309 323 273=0.006 0.00366professor Scimemi has accepted you as a Master’s student and you are involved in a project that studies the cellular basis of neuropsychiatric diseases. For your electrophysiology recordings you have to make a recording solution containing (in mM): 119 NaCl, 2.5 KCl, 2.5 CaCl2, 1 MgCl2, 26.2 NaHCO3, 1 NaH2PO4, 22 glucose. You need 800 ml of it. How much KCl do you need to weigh out? Hint: we only care about KCl here. (MW KCl is 74.55 g/mol1. C5H1002 10 HPM-00-297 100 TRANSMITERE 9 4000 8 T 3000 T 7 T 6 2000 5 ppm MOVENEDI T 4 3 T 1500 : T 3 1 T 2 : T 1000 6 T 1 T 0 500 200 DEPT-90 180 160 CDS-00-156 DEPT-135 140 120 100 ppm T 80 60 T 40 20 لند
- What is a 99% confidence interval uncertainty of the slope when we are given a trendline with a slope of 42.211 and an R² value of 0.9991 for a set of 145 data points?SPECTRA FOR HOMEWORK 11, CHE 230 002 This page is not to be submitted to Gradescope. Use these spectra as you answer questions on the Homework 11 document. Spectra for Problem 1 100 %T 80 60 40 20 Relative Intensity 4000 100- 80 8 60- 40- 20- 0 20 2H 2H 3000 m/z 50.0 75.0 76.0 155.0 157.0 183.0 185.0 212.0 214.0 100.0 97.5 11.8 11.7 1686 cm 40 60 80 2000 rel, intensity 11.9 16.9 17.6 28.8 27.8 Wavenumber[cm-1] 100 120 m/z 1500 7.90 7.85 7.80 7.75 7.70 7.65 7.60 7.55 1588 cm² 20 20 ₂ مسلسل..............للمسلسل ppm 140 160 180 200 220 2H 1000 3.12 3.00 2.95 2.90 3H 500 400 A. Propose a molecular formula for this compound B. Propuse. a structure selected 1.25 1.20 1.15 2011.4 A 6.25 x 10-³ M solution of potassium permanganate (KMnO4) has a transmittance of 47.4% when measured in a 1.5 cm cell at a wavelength of 625 nm. (a) Calculate the absorbance of the KMnO4 solution (b) Calculate the molar absorptivity of KMnO4





