
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
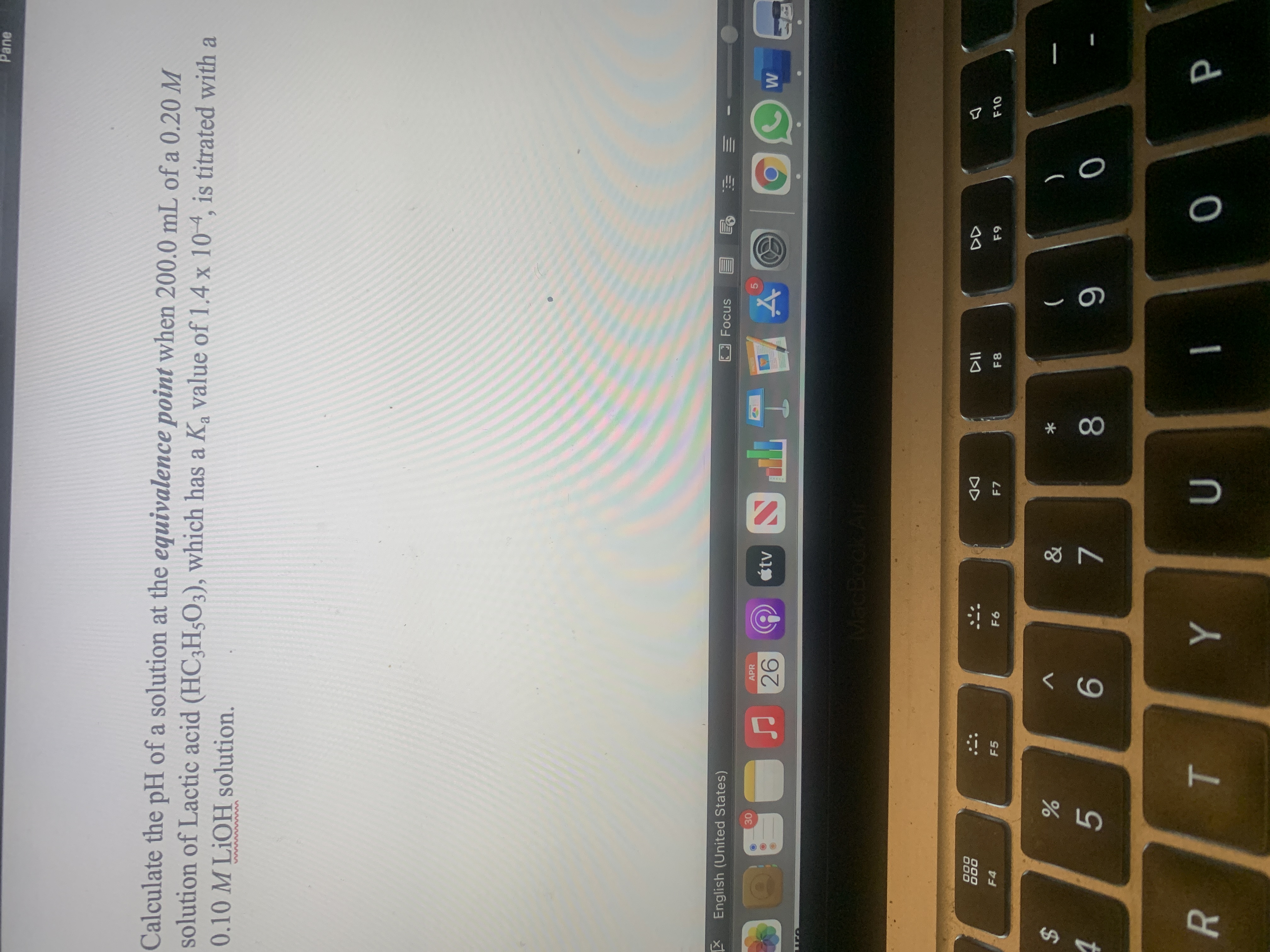
Transcribed Image Text:Calculate the pH of a solution at the equivalence point when 200.0 mL of a 0.20 M
solution of Lactic acid (HC;H;O3), which has a K value of 1.4 x 104, is titrated with a
6.
0.10 M LIOH solution.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine the pH during the titration of 59.5 mL of 0.490 M hypochlorous acid (Ką = 3.5×10-8) by 0.490 M NaOH at the following points. (Assume the titration is done at 25 °C.) (a) Before the addition of any NaOH (b) After the addition of 15.0 mL of NaOH (c) At the half-equivalence point (the titration midpoint) (d) At the equivalence point (e) After the addition of 89.3 mL of NaOHarrow_forwardAn analytical chemist is titrating 176.8 mL of a 0.7300M solution of butanoic acid (HC,H,CO,) with a 0.2000M solution of NaOH. The p K, of butanoic acid is 4.82. Calculate the pH of the acid solution after the chemist has added 770.3 mL of the NaOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of NaOH solution added. Round your answer to 2 decimal places. pH = Uarrow_forwardA buffer solution is made that is 0.415 M in CH3COOH and 0.415 M in CH3COO- . (1) If Ka for CH3COOH is 1.80×10-5 , what is the pH of the buffer solution? (2) Write the net ionic equation for the reaction that occurs when 0.103 mol HI is added to 1.00 L of the buffer solution. Use H3O+ instead of H+ .arrow_forward
- a An analytical chemist is titrating 86.1 mL of a 1.100M solution of hydrazoic acid (HN3) with a 0.7600M solution of KOH. The p K of hydrazoic acid is 4.72. Calculate the pH of the acid solution after the chemist has added 74.4 mL of the KOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of KOH solution added. Round your answer to 2 decimal places.arrow_forwardWhen 25.00 mL of 0.467 M NaOH was added to 50.00 mL of a 0.467 M solution of a weak acid, HX, the pH of the mixture reached a value of 3.49. What is the Ka of the weak acid? Ka=arrow_forwardAn analytical chemist is titrating 234.0 mL of a 0.4400M solution of butanoic acid (HC,H,CO,) with a 0.3900M solution of NaOH. The p K, of butanoic acid is 4.82. Calculate the pH of the acid solution after the chemist has added 63.61 mL of the NaOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of NaOH solution added. Round your answer to 2 decimal places.arrow_forward
- An analytical chemist is titrating 103.5 mL of a 0.3300M solution of acetic acid (HCH₂CO₂) with a 0.3600M solution of KOH. The p K of acetic acid is 4.70. Calculate the pH of the acid solution after the chemist has added 22.84 mL of the KOH solution to it. a Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of? KOH solution added. Round your answer to 2 decimal places. pH = Ś 000 18 Ararrow_forwardAn analytical chemist is titrating 240.2 mL of a 1.000M solution of acetic acid (HCH,CO, with a 0.2300M solution of KOH. The p K, of acetic acid is 4.70. Calculate the pH of the acid solution after the chemist has added 306.7 mL of the KOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of KOH solution added. Round your answer to 2 decimal places. pH =|arrow_forwardAn analytical chemist is titrating 145.0 mL of a 0.7900M solution of dimethylamine ((CH) NH) with a 0.8700M solution of HNO3. The p K, of dimethylamine is 3.27. Calculate the pH of the base solution after the chemist has added 93.9 mL of the HNO, solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HNO, solution added. Round your answer to 2 decimal places. pH =arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY