
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
thumb_up100%
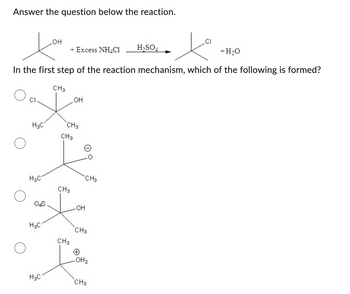
Transcribed Image Text:Answer the question below the reaction.
H3C
H3C
+ H₂O
In the first step of the reaction mechanism, which of the following is formed?
04S
H3C
OH
H₂C
+ Excess NH4C1
CH3
CH3
OH
CH3
CH3
CH3
-OH
CH3
CH3
(+)
.OH₂
H₂SO4
CH3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of these would be the product, represented by D, in the reaction sequence?arrow_forwardWhich of the following alkyl halides will react with Nal in acetone fastest to form a precipitate? O 1-fluoro-1-methylcyclopentane O 3-bromohexane O 2-chloro-2-methylpropane O 1-bromohexanearrow_forwardWhich of the following is an intermediate in the reaction below? A) H3t H₂CH H B (Sia)₂ ☆ó B) H₂ (~= UH CH₂CH H H E) H₂E NOH 1. (Sia) 2BH THF 2. NaOH, H₂ de. H D) H3 (1 нarrow_forward
- Place the best reagent and conditions in the bins for each of the following reactions. One of the bins require 2 choices, as indicated by the larger bin. (Stoichiometry is omitted.) OH NH₂ OL 2: OH منو 1. 2. CrO3 H₂O+ CH3NH₂ CH, ONa NaN3 Answer Bank CH3CO₂ Na NaCl OH LiAlH4 NH3 SOCI₂ NaBH4 ol iPrMgBr (CH3)2 CuLi CH, MgBr CH, OHarrow_forwardAtom mapping, the determination of the fate of atoms of a starting material in the product, can help in the construction of a reasonable mechanism. Using the following labeling scheme, determine which atoms in the starting material (which are labeled with numerals) correspond to the atoms in the product (which are labeled with letters). 2 3 11 12 a 8. H2SO4 10 13 7 OH d f Reactant Product Reactant Product 1 8. 9. 3 10 4 11 5 12 13 7 2.arrow_forwardUse the two-step mechanism below to answer the following question. Step 1: NO (g) + N2O (g) → N2 (g) + NO2 (g) Step 2: 2 NO2 (g)→2 NO (g) + O2 (g) What is the intermediate? O N20 O NO O N2 NO2 O 02 MacBook Air DII 80 888 F6 F7 FB F3 F4 & %23 %24 3 4 5 8 E R Y F C в M I >arrow_forward
- Please answer number 3carrow_forwardDraw the organic molecule(s) which is (are) formed in the following reaction. Do not include molecules like H2O or HCl. Please answer fast I give you upvotearrow_forwardNote: Solvents may be written following a comma. In this reaction, Et₂O is the solvent for step 1 and acetone the solvent for step 2. Solvents usually will not be directly involved the main reaction and are instead supplied for thoroughness. What is the major product formed in the following reaction? OA OB OC OD OE CN ai A CN B OH 1. PBrs, Et₂0 2. KCN, acetone -0 Br CN C Br CN D Br CN Earrow_forward
- 8. Draw a reaction coordinate diagram depicting the thermodynamics of the following reaction. Indicate the locations of any starting materials, intermediates, products, transition states, free energy of reaction and activation energy present in the diagram. Draw the transition state structure, complete with charge information, for the following transformation. Br H₂CCH₂H 20 CA CH3 H3CCH2OH + H CH3 CH3 + BI-arrow_forwardQUESTION 4 Use the reaction shown below to answer questions 4-5. HO Reaction C Reaction D Consider the structural changes that take place during Reaction C and Reaction D. (Make a list of objectives.) Which fundamental mechanisms take place during Reaction C and Reaction D? Choose one mechanism for each reaction. O Reaction C: SN2 O Reaction C: SN1 O Reaction C: E1 (Zaitzev's rule) OReaction D: E1 (Zaitzev's rule) O Reaction D: E2 (Hofmann's rule) O Reaction D: E2 (Zaitzev's rule) QUESTION 5 но Reaction C Reaction D Consider the structural changes that take place during Reaction C and Reaction D and the fundamental mechanisms that you identified from the previous question. Fill in the boxes with the missing reagent that is required to complete Reaction C and Reaction D? Choose one reagent per reaction. O Reaction C: HCI O Reaction C: Cl2, heat O Reaction C: Pyridine, SOCI2 O Reaction D. KOC(CH3)3 O Reaction D. NaOCH2CH3 O Reaction D: HOCH2CH3arrow_forwardSelect the correct product for this reaction. Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. a A b B C C d D E = 6 ? gy CHILOI Darrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY