
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
An experiment involves the rxn: 2HI(g) ⇄ H2(g) + I2(g).
A sample of pure HI was placed inside a rigid container at a certain temperature. The table provides the initial and equilibrium concentrations for some of the substances in the rxn. Based on the data, which of the following is the value of the equilibrium constant (Keq) for the reaction, and why?
- Keq= 2.5×10−1, because [I2]eq = 2×[HI]eq.
- Keq= 6.3×10−2, because [I2]eq = ½ [HI]eq.
- Keq= 1.6×10−2, because [I2]eq = [H2]eq.
- Keq= 3.1×10−2, because [I2]eq = 2×[H2]eq.
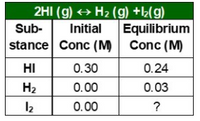
Transcribed Image Text:2HI (g) + H2 (g) +2(g)
Equilibrium
Conc (M)
Sub-
Initial
stance Conc (M)
HI
0.30
0.24
H2
0.00
0.03
12
0.00
?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A chemical engineer is studying the following reaction: HCH3CO2(aq)+CH3NH2(aq) → CH3CO2(aq) + CH3NH3(aq) At the temperature the engineer picks, the equilibrium constant K for this reaction is 0.54. The engineer charges ("fills") four reaction vessels with acetic acid and methylamine, and lets the reaction begin. She then measures the composition of the mixture inside each vessel from time to time. Her first set of measurements are shown in the table below. Predict the changes in the compositions the engineer should expect next time she measures the compositions. reaction vessel compound concentration expected change in concentration HCH,CO₂ 1.01 M O↑ increase CH3NH2 1.22 M O decrease O↑ increase O decrease O (no change) O (no change) A CH,CO, 0.64 M O↑ increase O decrease O (no change) CH, NH3 1.04 M O ↑ increase O decrease O (no change) HCH CO₂ 0.33 M O↑ increase O decrease O(no change) CHÍNH, 0.50 M O↑ increase O decrease O (no change) B CH,CO, 1.01 M O increase O decrease O (no…arrow_forwardA chemical engineer is studying the following reaction: BF3(aq) + NH3(aq) → BF3NH₂(aq) At the temperature the engineer picks, the equilibrium constant K for this reaction is 0.63. C The engineer charges ("fills") three reaction vessels with boron trifluoride and ammonia, and lets the reaction begin. He then measures the composition of the mixture inside each vessel from time to time. His first set of measurements are shown in the table below. Predict the changes in the compositions the engineer should expect next time he measures the compositions. reaction vessel A B C compound BF₂ NH₂ BF₂NH₂ BF 3 NH₂ BF3 NH3 BF 3 NH₂ BF₂NH3 concentration 0.47 M 0.48 M 1.03 M 0.46 M 0.47 M 1.04 M 0.94 M 0.95 M 0.56 M expected change in concent ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase O ↓ decrease ↓ decrease ↓ decrease ↓ decrease ↓ decrease ↓ decrease ↓ decrease ↓ decrease ↓decrease ation (no change) (no change) (no change) (no change) (no…arrow_forwardFor the chemical equation: SO2(g) + NO2(g) SO3(g) + NO(g) The equilibrium constant at a certain temperature is 8.80. At this temperature, calculate the number of moles of NO2(g) that must be added to 6.20 mol SO2(g) in order to form 4.40 mol SO3(g) at equilibrium.arrow_forward
- At a certain temperature, the equilibrium constant K for the following reaction is 352.: H,(g) + Cl,(g) 2HC1(g) Use this information to complete the following table. Suppose a 17. L reaction vessel is filled with 0.38 mol of HCI. What can you say about the composition of the mixture in the vessel at equilibrium? There will be very little H2 and Cl2 There will be very little HCI. Neither of the above is true. What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. K =0 2 HCl(g) H,(9)+Cl,(9) What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. K = | 2 H,(9)+2C12(9) 4 HCl(g)arrow_forwardMercury and oxygen react to form mercury(II) oxide, like this: 2 Hg(1)+O2(g)→2 HgO(5) At a certain temperature, a chemist finds that a 3.6 L reaction vessel containing a mixture of mercury, oxygen, and mercury(II) oxide at equilibrium has the following composition: compound amount Hg 9.7 g O2 21.8 g HgO 10.2 g Calculate the value of the equilibrium constant K̟ for this reaction. Round your answer to 2 significant digits. K_ = 0arrow_forwardThe equilibrium constant, K, for the following reaction is 3.92×10-2 at 531 K.PCl5(g) PCl3(g) + Cl2(g)An equilibrium mixture of the three gases in a 7.63 L container at 531 K contains 0.331 M PCl5, 0.114 M PCl3 and 0.114 M Cl2. What will be the concentrations of the three gases once equilibrium has been reestablished, if the volume of the container is increased to 18.0 L? [PCl5] = M [PCl3] = M [Cl2] = M Submit Answerarrow_forward
- At a certain temperature, the equilibrium constant K for the following reaction is 3.1 × 10 : H,(g) + I,(g) = 2 HI(g) Use this information to complete the following table. Suppose a 36. L reaction vessel is filled with 1.7 mol of HI. What can you say about the composition of the mixture in the vessel at equilibrium? There will be very little H2 and I2. There will be very little HI. Neither of the above is true. What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. K = 1 2 HI(g) H2(9)+I,(9) What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. K = I 2 H,(9)+21½(9) 4 HI(g)arrow_forwardMercury and oxygen react to form mercury(II) oxide, like this: 2 Hg(1)+O2(9)→2 HgO(s) At a certain temperature, a chemist finds that a 10. L reaction vessel containing a mixture of mercury, oxygen, and mercury(II) oxide at equilibrium has the following composition: compound amount Hg 12.7 g O2 13.4 g HgO 15.5 g Calculate the value of the equilibrium constant K for this reaction. Round your answer to 2 significant digits. K = | Submit Assignm Continue O 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center| Access 43.203 FEB 19 MacBook Airarrow_forwardWhen nitrogen gas reacts with oxygen gas, nitrogen monoxide gas forms: N2(g) + O2(g) ⇋2NO(g) Initially, 0.40 mol of each reactant is placed into a 2.0 L container and equilibrium is established. If Keq=50.1 for the reaction, what are the equilibrium concentrations of all reactants and products? What two conditions are required, if we want to move reaction forward.arrow_forward
- For the chemical equation SO, (g) + NO, (g) = So, (g) + NO(g) the equilibrium constant at a certain temperature is 2.70. At this temperature, calculate the number of moles of NO, (g) that must be added to 2.75 mol SO,(g) in order to form 1.10 mol SO, (g) at equilibrium. moles of NO,(g): molarrow_forwardA chem ngineer is studying the following reaction: + HCN(aq) + NH3(aq) → CN¯(aq)+NH (aq) At the temperature the engineer picks, the equilibrium constant K for this reaction is 1.1. C The engineer charges ("fills") four reaction vessels with hydrogen cyanide and ammonia, and lets the reaction begin. He then measures the composition of the mixture inside each vessel from time to time. His first set of measurements are shown in the table below. Predict the changes in the compositions the engineer should expect next time he measures the compositions. reaction vessel A B C compound HCN NH3 CN NH4 HCN NH3 CN NH₁ HCN NH₂ CN NHA 4 concentration 0.28 M 0.12 M 1.05 M 0.94 M 0.67 M 0.51 M 0.66 M 0.55 M 0.31 M 0.15 M 1.02 M 0.91 M expected change in concentration ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↑ increase ↓ decrease ↓decrease ↓ decrease 888888 ↓decrease ↓ decrease ↓decrease ↓ decrease ↓ decrease ↓decrease…arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY