
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
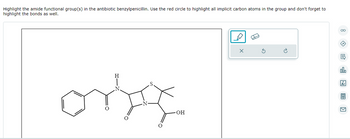
Transcribed Image Text:Highlight the amide functional group(s) in the antibiotic benzylpenicillin. Use the red circle to highlight all implicit carbon atoms in the group and don't forget to
highlight the bonds as well.
S
OH
X
民沁園图〗
Ar
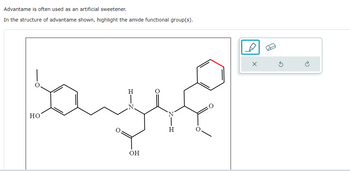
Transcribed Image Text:Advantame is often used as an artificial sweetener.
In the structure of advantame shown, highlight the amide functional group(s).
HO
H
OH
H
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Tertiary amides have how many C-N bonds?arrow_forwardClassify the specified amides as 1º, 2º, or 3º. Part 1 Select the single best answer. Part 2 Oxytocin, sold under the trade name Pitocin, is a naturally occurring hormone used to stimulate uterine contractions and induce labor. Classify the specified amides in oxytocin as 1º, 2º, or 3º.arrow_forwardQuestion 16.arrow_forward
- What is true regarding protein backbones? The protein backbone is different for each protein and contains both amine and carboxylic acid functional groups The protein backbone is the same for each protein and contains both amine and carboxylic acid functional groups The protein backbone is different for each protein and contains amide functional groups The protein backbone is the same for each protein and contains amide functional groupsarrow_forwardKindly give all the NEW IUPAC NAME and COMMON NAME of Roman Numeral I A, B, C. DRAW the STRUCTURES of the following on Roman Numeral II A, B, C, D Roman Numeral 3 give the reaction of the given amidesarrow_forward2. Draw the product when each of the amines acts as a base when combined with water. Name the ion that forms. a) CH-CHL- Nith + Hy0 b) CHy-NH-CH CHy + HgOarrow_forward
- This compound is mescaline, a hallucinogen. CHO CHO T Which of the following statements is true? Select one: O a. O b. O c. O d. Mescaline is an amine. Mescaline contains an alcohol group. Mescaline is a carboxylic acid. Mescaline is an amine and contains an alcohol group.arrow_forwardquestion 7arrow_forwardQUESTION 3 Which of the following best describes the reaction below. I H₂C NHCH3 H₂O*, heat H₂C OH O The reaction is incorrect, the amine should form along withe the carboxylate because an acid was used. O The reaction is incorrect, acids can not break amines into carboxylic acids and amides. The reaction is correct, the protonated amine forms because an acid is used to break the amide. O The reaction is incorrect, adding acids to amides forms ketones and amines, not carboxylic acids. +NH,CH*arrow_forward
- Which represents the acid form of a primary amine? R-NH₂ R-NH R-NH, OR-NH3arrow_forwardDraw the structure of the following three isomeric amides with chemical formula C6H11NO. Amide #1: (E)-N,N-dimethyl-2-butenamide Amide #2: (Z)-N-methyl-3-pentenamide Amide #3: (Z)-3-hexenamidearrow_forwardThe product of this reaction is: O CI secondary amide secondary amine primary amide tertiary amide primary amine + 2NH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY