
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
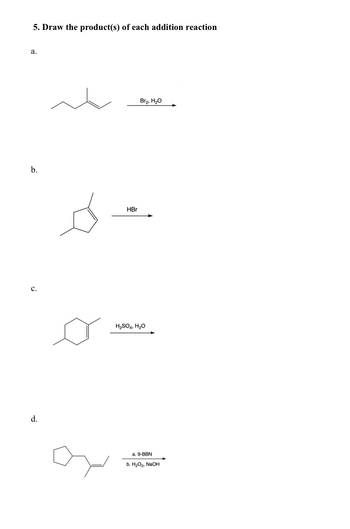
Transcribed Image Text:a.
5. Draw the product(s) of each addition reaction
b.
d.
دھ
HBr
H2SO4, H₂O
Bra, H2O
a. 9-BBN
b. H2O2, NaOH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 1 steps with 2 images

Knowledge Booster
Similar questions
- 30. What is the IUPAC name for this compound? CH3 0 1 I CH3-CH-CH₂-C-OH A. pentanoic acid B. 3-methylbutanoic acid C. 2-methylbutanoic acid D. 2-methyl-4-butanoic acidarrow_forwardName the alkyl halide below CI a. 4-chlorohexane O b. chlorohexane O c. 1-ethyl-1-chlorobutane O d. 3-chlorohexanearrow_forwardGive the IUPAC name for each. a. I have 5-bromo-3methylheptanearrow_forward
- Br. 21. CH3 H CH; The compound above can exhibit a. cis-isomerism only O b. trans-isomerism only c. both cis- and trans-isomerism O d. neither cis- nor trans-isomerismarrow_forwardReaction A. NaOEt EIOH Incorrect Reaction B. (CH3,COK (CH3,COH Incorrectarrow_forwardIf an alkyne has 10 hydrogen atoms with a single triple bond, how many carbon are in the structure? a. 4 b. 5 c. 6 d. 7arrow_forward
- 10. How many H-bonds can a secondary alcohol form? A. 3 B. 1 C. 2 D. 0arrow_forward10.The reaction of excess Grignard reagent with an ester of formic acid, HCO2R, gives a. methanol. b.a tertiary alcohol. c. primary alcohol d.secondary alcohol.arrow_forwardWhich is the major organic product of this reaction? A. B. 4 ОА O OB O C COD AICI3 benzene C. D.arrow_forward
- What is the IUPAC name for the following 2. What is the IUPAC name for the compound? following compound? u 1. ГОН 2-methylpentanoic acid 3-methylpentanoic acid 2-methylhexanoic acid 3-methylhexanoic acid None of these What is the IUPAC name for the following 4. compound? A. B. C. D. 3. A. B. C. D. E. sec-butyl ethanoate ethyl 3-methylpentanoate 3-methylbutyl ethanoate ethyl 3-methylbutanoate none of these A BUDE A. B. C. A. B. ABCDE What is the IUPAC name for the following compound? CI C. D. E. a-methylbutyryl chloride B-methylbutyryl chloride y-methylbutyryl chloride 2-methylbutanoyl chloride 3-methylbutanoyl chloride propanoic anhydride butanoic anhydride propionic anhydride pentanoic anhydride butyric anhydridearrow_forward62. What is the product of this reaction? 1) H. of H. 2) H* C d. OH O H. H.arrow_forward3. Choose the correct product of the dehydration of 4,4-dimethyl-3-hexanol(draw out the reaction). A. 4,4-dimethyl-2-hexene B. 3,4-dimethyl-2-hexene C. 4,4-dimethyl-1-hexene D. 3,3-dimethyl-4-hexene A B.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY