
College Physics
11th Edition
ISBN: 9781305952300
Author: Raymond A. Serway, Chris Vuille
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
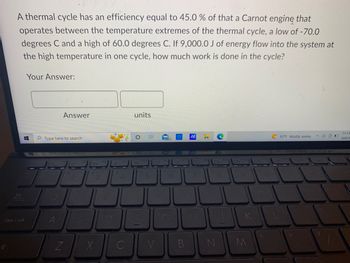
Transcribed Image Text:A thermal cycle has an efficiency equal to 45.0 % of that a Carnot engine that
operates between the temperature extremes of the thermal cycle, a low of -70.0
degrees C and a high of 60.0 degrees C. If 9,000.0 J of energy flow into the system at
the high temperature in one cycle, how much work is done in the cycle?
HHH
Kimamey
Your Answer:
Caps Lock
Type here to search
Answer
JANG UGLE (O 260LCH
A
N
7
3
X
C
4
C
units
O
5
99+
113
V
B
8
N
K
M
C83°F Mostly sunny
l
11:13
8/8/2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Heat of amount 250 J is removed from a heat reservoir at a temperature of 270 K. What is the entropy change of the reservoir? Hint ΔS=ΔS= J/KJ/K. (Use the sign to indicate whether entropy of reservoir increases or decreases.) Submit QuestionQuestion 9 It is found that an engine discharges 50 J while absorbing 85 J each cycle of operation. Hint What is the efficiency of the engine? eff=eff= How much work does it perform per cycle? W=W= Jarrow_forwardRefrigeration Cycle Operating As Shown In The Figure Below Has A Coefficient Of Performance Β = 1.8. For The Cycle, Qout = 250 kj. Determine Qin and Wcycle, each in kJarrow_forwardFor a power cycle operating as shown in the figure, Wcycle = 800 Btu and Qout = 1800 Btu. Hot body System Cold body Cout W cycle-in-out What is the heat input, Qin, in Btu, and what is the cycle thermal efficiency?arrow_forward
- Imagine that a high-efficiency heat engine could be created that operated betweeen the lot and high temperatures of -50.5 degrees celsius and 5550 degrees celsius. What would be the maximum efficiency of such an engine?arrow_forwardHeat engine: efficiency 19% , work 1510J How much heat is given off to the cold reservoir ?? In Jarrow_forwardNeeds Complete typed solution with 100 % accuracy.arrow_forward
- Please how to solve this questionsarrow_forwardA carnot enine removes 1200J of heat from a high temperature source and dumps 450J to the atmosphere at18.0C What is the efficiency of the engine?What is the temperature of thehot reservoir?arrow_forwardSuppose a heat engine design makes a square on a PV diagram, using a monatomic gas. The high pressure is 3P and low pressure is P. The high volume is 3V and low volume is V. What is the efficiency of the heat engine?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

University Physics (14th Edition)
Physics
ISBN:9780133969290
Author:Hugh D. Young, Roger A. Freedman
Publisher:PEARSON

Introduction To Quantum Mechanics
Physics
ISBN:9781107189638
Author:Griffiths, David J., Schroeter, Darrell F.
Publisher:Cambridge University Press

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Lecture- Tutorials for Introductory Astronomy
Physics
ISBN:9780321820464
Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina Brissenden
Publisher:Addison-Wesley

College Physics: A Strategic Approach (4th Editio...
Physics
ISBN:9780134609034
Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart Field
Publisher:PEARSON