
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
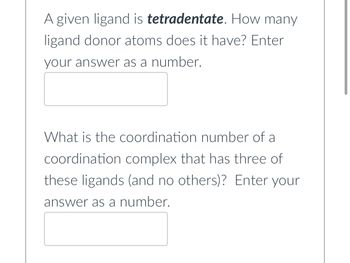
Transcribed Image Text:A given ligand is tetradentate. How many
ligand donor atoms does it have? Enter
your answer as a number.
What is the coordination number of a
coordination complex that has three of
these ligands (and no others)? Enter your
answer as a number.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps with 16 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following statements is not true? A. Coordination Chemistry is the science concerned with the interactions of organic and inorganic ligands with metal centers. B. Coordination compounds are composed of a metal atom or ion and one or more ligands (atoms, ions, or molecules) that donate electrons to the metal. C. Coordination compounds include organometallic compounds. D. The modern theory of coordination chemistry is based largely on the work of Alfred Werner. E. Coordination compounds are examples of Brønsted-Lowry acid-base adducts.arrow_forwardDimethyldithiocarbamate (abbreviated as dmdtc) is an anionic bidentate ligand with a chemical formula (S2CNMe2)- - where Me stands for methyl group (-CH3). In the photo above, this ligand coordinates to a metal through its 2 sulfur atoms (where M represents a metal). When three dmdtc ligands bind to a ferric ion (Fe3+), a stable coordination complex called ferbam is formed. Given this information, answer / identify the following. complex geometry hybridization type of the central atom coordination numberarrow_forwardConsider this metal complex: Br Br Cl C1 8 \. Br Complex A Now decide whether each complex in the table below is another example of A, an isomer of A, or an entirely different chemical compound. Complex Br Br Br Br 8 J J J Cl Br 8 8 V C1 Br 8 Cl Br 5 J 2 Br 3- This complex is ... O the same as A ... an isomer of A ... a different compound. ...the same as A O an isomer of A ... a different compound. O ... the same as A ...an isomer of A O a different compound. O ... the same as A O ... an isomer of A ... a different compound. Marrow_forward
- Consider the coordination complex [Cr(OH₂)3C13] Br3 Identify the ligands by name (include the word "ion" at the end for all ions): What is the charge on the coordination complex? Enter the sign then the numeric value (+2, -2, etc.) What is the oxidation state of the Cr ion? Enter the sign then the numeric value (+2, -2, etc.) What is the coordination number for the complex ion? Enter your answer as a number.arrow_forwardWhich statements are true regarding ligands? Select all that apply. Ligands donate all electrons involved in bonds they form with metal cations. Ligands are electron pair acceptors in coordination complexes. Ligands act as Lewis bases in coordination complexes. Ligands form ionic bonds with transition metals. All ligands with two lone pairs are bidentate. A ligand that can donate more than one electron pair to a metal cation is called a chelate. One ligand can donate only one lone pair to a metal cation.arrow_forwardYou are comparing two coordination complexes that have the same molecular shape and central metal cation. Complex A has only chloride ligands and Complex B had only CO ligands. Which statements are true regarding the complexes? Select all that apply. Complex A has the smaller crystal field splitting. Light of higher frequency is required to promote an electron from the lower to the higher d orbitals in complex A relative to complex B. The light emitted as a photon is released to relax an electron from higher to lower energy d orbitals has a longer wavelength for complex A. Light of higher frequency is required to promote an electron from the lower to the higher d orbitals in complex B relative to complex A. The light emitted as a photon is released to relax an electron from higher to lower energy d orbitals has a longer wavelength for complex B. Complex B has the smaller crystal field splitting.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY